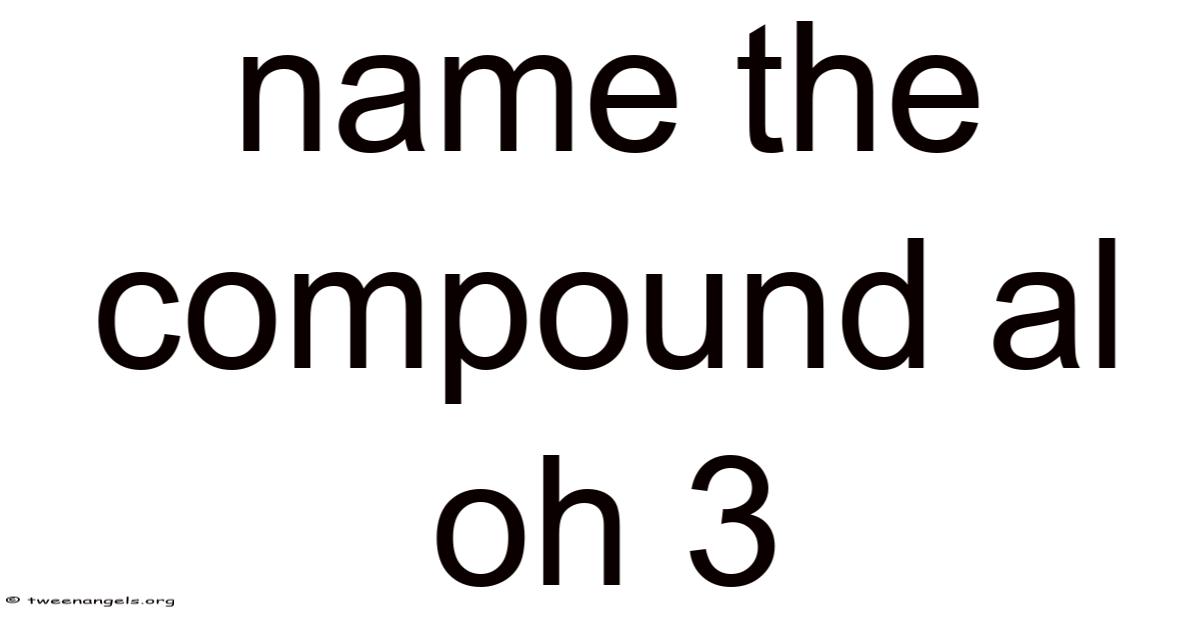Al(OH)₃: Understanding the Chemistry, Uses, and Everyday Significance of Aluminum Hydroxide
Aluminum hydroxide, with the chemical formula Al(OH)₃, is a compound that quietly plays a important role in many industrial processes, everyday household products, and even biological systems. Despite its modest appearance—a white, powdery solid that dissolves only in strongly acidic or basic solutions—Al(OH)₃ is a fascinating example of how a simple arrangement of atoms can have wide-reaching implications. In this article we’ll explore its structure, how it’s named, its key applications, safety considerations, and some frequently asked questions Which is the point..
Introduction
When you hear “Al(OH)₃,” you might think of a laboratory reagent or a component of antacid tablets. In reality, this compound is a cornerstone in water treatment, pharmaceuticals, ceramics, and even food preservation. Understanding why it’s called aluminum hydroxide and how it behaves chemically provides insight into its versatility and importance.
Naming the Compound: From Formula to Common Name
1. Systematic Naming (IUPAC)
The International Union of Pure and Applied Chemistry (IUPAC) prescribes systematic names based on the elemental composition:
- Al stands for the metal aluminum.
- (OH)₃ indicates three hydroxide groups attached to the aluminum atom.
Thus, the systematic name is aluminum hydroxide. The formula Al(OH)₃ is a concise representation that conveys the same information: one aluminum atom coordinated to three hydroxide ions That's the part that actually makes a difference..
2. Common Names and Synonyms
- Aluminia (especially in the context of its oxide form, Al₂O₃, after dehydration)
- Aluminium hydroxide (British spelling)
- Aluminum hydroxide (American spelling)
These synonyms often appear in literature, product labels, and regulatory documents.
3. Naming in Context
In aqueous solutions, Al(OH)₃ is often referred to as aluminum hydroxide precipitate or aluminum hydroxide gel, reflecting its tendency to form a gelatinous suspension when added to water or mildly acidic solutions.
Structural and Physical Characteristics
| Property | Value | Notes |
|---|---|---|
| Formula | Al(OH)₃ | |
| Molar mass | 78.00 g/mol | |
| Appearance | White, fine powder | |
| Solubility | Insoluble in water | Dissolves in strong acids or bases |
| pH of saturated solution | ~7 | Slightly alkaline due to surface hydroxyl groups |
| Crystal structure | Trigonal, corundum-like | Similar to α‑Al₂O₃ in dehydrated form |
The official docs gloss over this. That's a mistake.
Al(OH)₃ crystallizes in a trigonal lattice where each aluminum is octahedrally coordinated by six oxygen atoms (three from hydroxide groups and three from neighboring hydroxides). This arrangement gives the compound a relatively high hardness and makes it useful as a refractory material.
Chemical Behavior and Reactions
1. Hydrolysis and Precipitation
When a soluble aluminum salt (e.g., AlCl₃) is introduced into water, the following equilibrium occurs:
[ \text{Al}^{3+} + 3\text{OH}^- \rightleftharpoons \text{Al(OH)}_3(s) ]
Because Al(OH)₃ is poorly soluble, it precipitates out as a gelatinous solid, which can be filtered or centrifuged Still holds up..
2. Acid–Base Equilibria
Al(OH)₃ acts as a weak base:
[ \text{Al(OH)}_3 + 3\text{H}^+ \rightarrow \text{Al}^{3+} + 3\text{H}_2\text{O} ]
In strongly acidic media, the hydroxide groups are protonated, leading to dissolution.
3. Dehydration to Alumina
Upon heating to temperatures above 300 °C, Al(OH)₃ dehydrates to form alumina (Al₂O₃):
[ 2\text{Al(OH)}_3 \xrightarrow{\Delta} \text{Al}_2\text{O}_3 + 3\text{H}_2\text{O} ]
This process is exploited in the production of high‑purity alumina used in ceramics and electronics The details matter here. Practical, not theoretical..
Industrial and Commercial Applications
| Application | Role of Al(OH)₃ | Key Benefit |
|---|---|---|
| Antacids | Neutralizes excess stomach acid | Rapid relief from heartburn |
| Water treatment | Flocculant and coagulant | Removes heavy metals, dyes, and pathogens |
| Catalyst support | Provides high surface area | Enhances catalytic activity in petrochemical processes |
| Fire retardant | Releases water vapor upon heating | Reduces flammability of polymers |
| Ceramics & refractories | Forms a stable, heat‑resistant matrix | Improves strength and thermal shock resistance |
| Food additive | Acts as a pH regulator | Stabilizes acidic foods and beverages |
| Pharmaceutical excipient | Fills tablets, controls dissolution | Improves drug delivery and stability |
The versatility of Al(OH)₃ stems from its amphoteric nature: it can act as both an acid and a base, allowing it to interact with a wide range of substances That's the part that actually makes a difference. No workaround needed..
Safety and Environmental Considerations
1. Human Health
- Inhalation: Fine dust can irritate the respiratory tract. Protective masks are recommended in industrial settings.
- Skin and Eye Contact: Generally mild irritation; washing with water usually suffices.
- Ingestion: Small amounts are harmless; large doses can cause gastrointestinal upset.
2. Environmental Impact
- Biodegradability: Al(OH)₃ is not readily biodegradable but is considered low toxicity to aquatic life.
- Accumulation: In large quantities, it can contribute to increased alkalinity in water bodies, potentially affecting local ecosystems.
3. Regulatory Status
- Food-grade: Approved as a food additive (E number: E504) for use as a pH stabilizer.
- Pharmaceutical: Classified as a pharmaceutical excipient with established safety profiles.
Frequently Asked Questions (FAQ)
| Question | Answer |
|---|---|
| **What is the difference between Al(OH)₃ and Al₂O₃?Even so, ** | It is used as an antacid but not as a nutritional supplement. |
| **What happens if Al(OH)₃ is exposed to high heat?On top of that, | |
| **Is aluminum hydroxide safe for use in cosmetics? | |
| **Does Al(OH)₃ dissolve in normal water?And ** | Yes, it is used as a mild abrasive and skin conditioner; however, formulations must comply with cosmetic regulations. |
| Can Al(OH)₃ be used as a dietary supplement? | Al(OH)₃ is the hydrated form; Al₂O₃ (alumina) is the dehydrated, crystalline oxide obtained by heating. ** |
Conclusion
Aluminum hydroxide (Al(OH)₃) exemplifies how a seemingly simple inorganic compound can intersect multiple facets of science and industry. From neutralizing stomach acid to strengthening ceramics and purifying water, its applications are diverse and indispensable. Because of that, understanding its naming conventions, structural properties, and chemical behavior not only satisfies academic curiosity but also empowers professionals across chemistry, engineering, and health sciences to harness its full potential responsibly. Whether you’re a student, a researcher, or a curious reader, appreciating the role of Al(OH)₃ enriches your perspective on the hidden chemistry that shapes our everyday world.
Conclusion
Aluminum hydroxide (Al(OH)₃) exemplifies how a seemingly simple inorganic compound can intersect multiple facets of science and industry. From neutralizing stomach acid to strengthening ceramics and purifying water, its applications are diverse and indispensable. Continued research into enhancing its biodegradability and exploring sustainable production methods will further solidify Al(OH)₃’s position as a valuable material while minimizing any adverse consequences. Understanding its naming conventions, structural properties, and chemical behavior not only satisfies academic curiosity but also empowers professionals across chemistry, engineering, and health sciences to harness its full potential responsibly. Despite its widespread utility, careful consideration of its environmental impact – particularly regarding potential alkalinity shifts in aquatic ecosystems – remains crucial. Think about it: whether you’re a student, a researcher, or a curious reader, appreciating the role of Al(OH)₃ enriches your perspective on the hidden chemistry that shapes our everyday world. The bottom line: the continued study and responsible application of this versatile compound promise to yield even more innovative solutions across a broad spectrum of fields, demonstrating the enduring significance of basic inorganic chemistry.