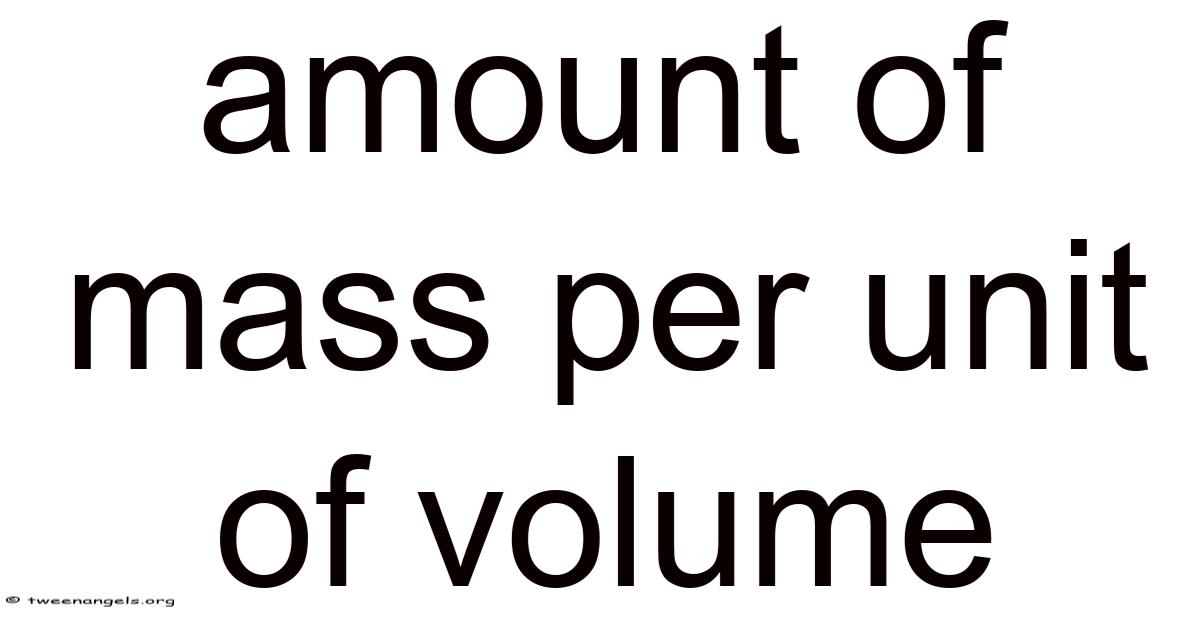Amountof mass per unit of volume is a fundamental physical quantity that describes how much matter is packed into a given space. In scientific terminology this concept is known as density, and it serves as the cornerstone for fields ranging from engineering and chemistry to geology and meteorology. Understanding how to measure, calculate, and interpret this property enables students and professionals alike to predict material behavior, design efficient systems, and solve real‑world problems with confidence.
Introduction
The amount of mass per unit of volume quantifies the compactness of a substance. This simple ratio carries profound implications: a high value indicates that the material is heavy for its size, while a low value suggests it is light and airy. Consider this: everyday examples abound—air feels “light” because its density is low, whereas lead feels “heavy” because its density is high. Even so, when you divide the total mass of an object by the space it occupies, the result is its density. By mastering this concept, learners can transition from abstract theory to practical application, making informed decisions in everything from cooking to spacecraft design.
How to Calculate It ### Basic Formula
The calculation follows a straightforward mathematical expression:
-
Measure the mass of the object using a calibrated scale.
-
Determine the volume occupied by the object, typically in cubic meters (m³) or cubic centimeters (cm³).
-
Apply the formula:
[ \text{Density} = \frac{\text{Mass}}{\text{Volume}} ]
The resulting unit is often expressed as kilograms per cubic meter (kg/m³) or grams per cubic centimeter (g/cm³).
Practical Steps
- Select appropriate instruments: Use a digital balance for precise mass measurement and a graduated cylinder, pycnometer, or laser scanner for volume assessment.
- Account for temperature and pressure: These variables can alter volume, especially for gases, so record environmental conditions.
- Convert units consistently: make sure mass and volume share compatible units before division to avoid errors.
- Repeat measurements: Conduct multiple trials and calculate an average to improve accuracy and reduce random error.
Example Calculation
Suppose a metal block has a mass of 250 g and a volume of 100 cm³.
[ \text{Density} = \frac{250\ \text{g}}{100\ \text{cm}^3} = 2.5\ \text{g/cm}^3 ]
This value places the material among moderately dense metals, such as aluminum.
Scientific Explanation
Why Density Matters
Density emerges from the interplay between mass—the amount of matter—and volume—the space that matter occupies. At the molecular level, particles are arranged differently depending on the substance: tightly packed arrangements yield high density, while loosely arranged structures produce low density. This arrangement is influenced by factors such as intermolecular forces, temperature, and pressure.
States of Matter
- Solids: Particles are tightly bound in a fixed lattice, resulting in high density.
- Liquids: Particles retain close proximity but can flow, leading to moderate density.
- Gases: Particles are widely spaced, giving gases the lowest densities under ordinary conditions.
Temperature and Pressure Effects
- Gases: According to the ideal gas law (PV = nRT), increasing temperature expands volume, thereby decreasing density, while increasing pressure compresses the gas, raising density.
- Solids and Liquids: Thermal expansion can slightly increase volume, causing a modest decrease in density; however, the effect is generally smaller than for gases.
Specific Gravity A related concept is specific gravity, which compares the density of a substance to that of a reference material—commonly water at 4 °C (1 g/cm³). Specific gravity is dimensionless and useful for quickly assessing whether a material will float or sink in water.
FAQ
What units are commonly used for density?
The International System of Units (SI) expresses density in kilograms per cubic meter (kg/m³). In laboratory settings, grams per cubic centimeter (g/cm³) or kilograms per liter (kg/L) are also prevalent And that's really what it comes down to..
Can density be negative?
No. Density is a ratio of two positive quantities (mass and volume), so it is always non‑negative. That said, in specialized contexts like negative mass in certain cosmological models, the term may appear, but it does not apply to ordinary materials.
How does density affect buoyancy?
An object will float in a fluid if its average density is lower than the fluid’s density; conversely, it will sink if its density is higher. This principle underlies ship design, hot‑air balloons, and the behavior of oil spills in water.
Why does ice float on water?
Ice has a lower density (≈0.917 g/cm³) than liquid water (≈1.000 g/cm³) because its crystalline structure creates open spaces. This rarity is why ice forms a surface layer that insulates aquatic life during winter.
Is density constant for all samples of the same material?
Not necessarily. Impurities, temperature variations, and
FAQ (continued):
Is density constant for all samples of the same material?
Not necessarily. Impurities, temperature variations, and differences in material composition or processing methods can lead to variations in density. Here's one way to look at it: a pure metal sample will have a consistent density, but impurities or defects in its structure may reduce it. Similarly, thermal expansion or compression can temporarily alter density, even within the same sample under changing conditions.
Conclusion
Density is a fundamental property that reflects how matter is organized at the microscopic level. From the tightly packed atoms in solids to the dispersed particles in gases, density determines how substances interact with their environment, influencing everything from buoyancy and material strength to natural processes like weather patterns and ocean currents. Its study bridges physics, chemistry, engineering, and environmental science, offering insights into both the microscopic world and large-scale phenomena. By understanding density, we gain tools to predict material behavior, optimize industrial processes, and address challenges ranging from sustainable design to climate resilience. When all is said and done, density is not just a measure of mass per volume—it is a key to unlocking the behavior and properties of the physical world around us Easy to understand, harder to ignore..