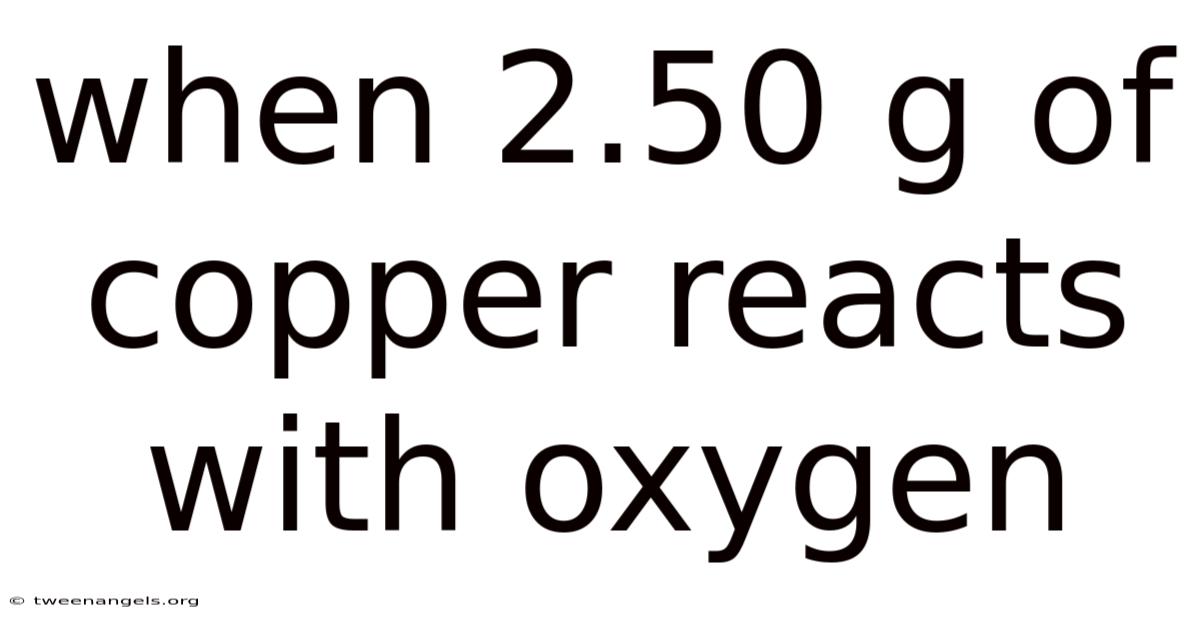When2.Which means 50 g of copper reacts with oxygen, a fundamental chemical process occurs that illustrates the principles of oxidation and stoichiometry. This reaction is not only a classic example of a metal reacting with a nonmetal but also a practical demonstration of how mass and molar relationships govern chemical transformations. In practice, the interaction between copper and oxygen is a cornerstone of understanding redox reactions, which are critical in both natural and industrial contexts. By examining this specific scenario, we can explore the underlying chemistry, the conditions required for the reaction to proceed, and the measurable outcomes that result from the interaction of these two elements.
The chemical reaction between copper and oxygen is a well-documented process that typically requires heating to initiate. The balanced chemical equation for this reaction is 2Cu + O₂ → 2CuO. Day to day, this equation indicates that two moles of copper react with one mole of oxygen gas to produce two moles of copper(II) oxide. On top of that, when copper metal is exposed to oxygen in the air, especially under elevated temperatures, it undergoes oxidation to form copper oxide. The formation of copper oxide is a key outcome of this reaction, and its properties—such as its black color and solid state—contrast sharply with the reddish-brown appearance of pure copper. This visual change is a clear indicator of the chemical transformation taking place.
To understand the specifics of the reaction involving 2.50 g of copper, Make sure you calculate the quantities of reactants and products involved. It matters. Day to day, first, the molar mass of copper (Cu) is approximately 63. 55 g/mol. In practice, using this value, 2. 50 g of copper corresponds to 2.Also, 50 g ÷ 63. 55 g/mol ≈ 0.0393 moles of copper. In practice, according to the balanced equation, two moles of copper react with one mole of oxygen. So, 0.Now, 0393 moles of copper would require 0. 01965 moles of oxygen gas. The molar mass of oxygen (O₂) is 32 g/mol, so the mass of oxygen needed is 0.Worth adding: 01965 mol × 32 g/mol ≈ 0. 629 g. This calculation highlights the stoichiometric relationship between the reactants.
The formation of copper(II)oxide is not only a visual transformation but also a testament to the predictability of chemical reactions governed by stoichiometric principles. The calculated mass of 3.Worth adding: 13 g of CuO aligns with the law of conservation of mass, as the total mass of reactants (2. 50 g Cu + 0.On the flip side, 629 g O₂) equals approximately 3. 13 g of product. This consistency reinforces the reliability of balanced chemical equations in predicting outcomes. Adding to this, the reaction’s dependence on temperature highlights the role of energy in driving redox processes. In real terms, while the reaction can occur slowly at ambient conditions, heating significantly accelerates it by providing the activation energy needed to break bonds in copper and oxygen molecules. This interplay between energy and reaction kinetics is a fundamental concept in thermodynamics and industrial chemistry, where controlled reactions are essential for processes like metal refining or oxide synthesis Easy to understand, harder to ignore. Turns out it matters..
The broader implications of this reaction extend beyond the laboratory. Worth adding: copper oxide, for instance, is utilized in applications such as pigments, catalysts, and even in the production of semiconductors. Still, its ability to conduct electricity under certain conditions also makes it relevant in electronic materials. Additionally, the oxidation of metals like copper is a natural process in corrosion, underscoring the importance of understanding such reactions to prevent material degradation in infrastructure and electronics. By mastering the stoichiometry and mechanisms of this reaction, chemists can design more efficient industrial processes and develop materials with tailored properties.
At the end of the day, the reaction between copper and oxygen exemplifies the elegance of chemical stoichiometry and redox chemistry. On top of that, it serves as a foundational example for understanding how mass relationships and electron transfer govern chemical transformations. Beyond theoretical education, this reaction has practical relevance in materials science, environmental management, and technology. Mastery of such principles not only deepens our comprehension of chemical behavior but also empowers innovation across scientific and engineering disciplines. The simplicity of this reaction belies its profound impact, illustrating how basic chemical principles underpin both natural phenomena and human-made advancements.
At the end of the day, the reaction between copper and oxygen exemplifies the elegance of chemical stoichiometry and redox chemistry. In practice, it serves as a foundational example for understanding how mass relationships and electron transfer govern chemical transformations. Mastery of such principles not only deepens our comprehension of chemical behavior but also empowers innovation across scientific and engineering disciplines. Beyond theoretical education, this reaction has practical relevance in materials science, environmental management, and technology. The simplicity of this reaction belies its profound impact, illustrating how basic chemical principles underpin both natural phenomena and human-made advancements The details matter here. And it works..
Adding to this, the color change – transitioning from a silvery-blue to a reddish-brown – is a readily observable indicator of the reaction’s progress, offering a simple yet effective method for monitoring oxidation. Because of that, this visual cue is frequently employed in educational demonstrations and even in rudimentary analytical techniques. The formation of copper(I) oxide (Cu₂O) and copper(II) oxide (CuO) represents a shift in oxidation states for the copper atom, a core concept in understanding redox reactions. These different oxide forms possess distinct physical and chemical properties, contributing to the diverse applications of copper oxides The details matter here. Practical, not theoretical..
Delving deeper, the reaction’s rate isn’t solely dependent on temperature; factors like surface area of the copper, the partial pressure of oxygen, and the presence of catalysts can all significantly influence the speed of oxidation. Practically speaking, for instance, finely divided copper powder will oxidize much faster than a solid copper sheet due to its increased surface area exposed to the reacting oxygen. Similarly, introducing a catalyst, such as carbon, can lower the activation energy and accelerate the process. Understanding these nuances allows for precise control over the oxidation process, crucial in industries seeking to produce specific copper oxide grades It's one of those things that adds up..
People argue about this. Here's where I land on it.
Looking ahead, research continues to explore novel methods for manipulating this reaction, including utilizing plasma techniques to achieve rapid and uniform oxidation. Scientists are also investigating the potential of copper oxide nanoparticles in advanced applications like solar cells and sensors, capitalizing on their unique electronic and optical properties. The ongoing investigation into the reaction’s intricacies promises to yield further advancements in materials science and contribute to more sustainable and efficient industrial practices That's the part that actually makes a difference..
So, to summarize, the reaction between copper and oxygen represents far more than a simple chemical transformation. Plus, it’s a microcosm of fundamental chemical principles, demonstrating the interplay of thermodynamics, kinetics, and stoichiometry. From its historical significance in metallurgy to its burgeoning role in modern technology, this seemingly straightforward reaction continues to provide valuable insights and inspire innovation, solidifying its place as a cornerstone of chemical understanding and a testament to the power of basic scientific principles.
The underlying dynamics of this reaction extend beyond the laboratory, influencing both natural systems and technological innovations. Its ability to reveal oxidation states and surface characteristics makes it an essential tool for scientists seeking clarity in complex chemical systems. As research progresses, the interplay between these elements becomes even more apparent, underscoring the importance of such studies in refining industrial processes.
Also worth noting, the evolving techniques to optimize this transformation highlight our growing mastery over material properties. On top of that, by tailoring variables like temperature, surface structure, and reactant ratios, researchers can fine-tune outcomes that are vital for producing high-quality copper oxides. This precision not only enhances efficiency but also opens doors to new applications in advanced fields It's one of those things that adds up..
This reaction exemplifies the synergy between fundamental science and practical application, reminding us of the broader impact of even the most basic chemical interactions. As we continue to explore and innovate, the lessons drawn from these processes will undoubtedly shape the future of technology and sustainability That's the part that actually makes a difference. Still holds up..
Simply put, the copper oxidation reaction serves as a compelling testament to the interconnectedness of chemistry, engineering, and discovery. Its significance extends beyond the moment of observation, reinforcing the value of curiosity and perseverance in scientific advancement. The journey through this reaction underscores how foundational concepts can inspire transformative progress And that's really what it comes down to..
Short version: it depends. Long version — keep reading.