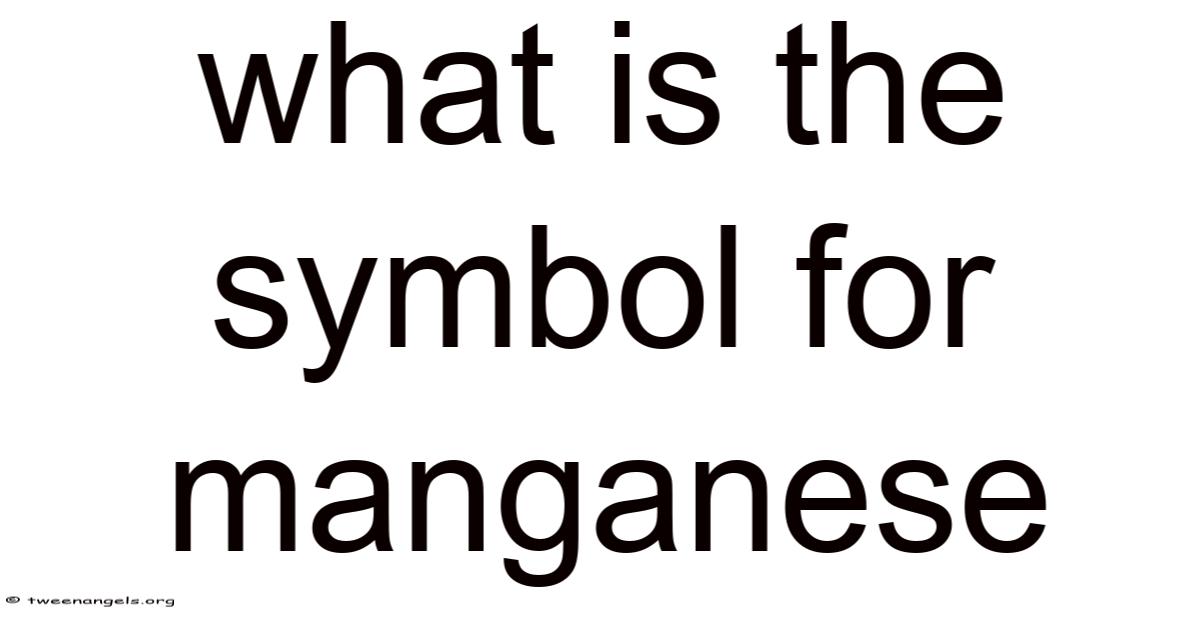What Is the Symbol forManganese? A Deep Dive into Its Significance and Origins
The symbol for manganese is Mn, a concise yet powerful representation of one of the most versatile elements in the periodic table. This two-letter abbreviation might seem arbitrary to some, but it carries a rich history, scientific precision, and practical relevance. Understanding why manganese is represented by Mn requires exploring its origins, chemical properties, and the role it plays in both natural and industrial contexts No workaround needed..
Short version: it depends. Long version — keep reading.
The Origins of the Symbol: A Historical Perspective
The symbol Mn for manganese is rooted in its Latin name, Manganum. This name itself has an intriguing backstory. Also, the element was first isolated in 1774 by the Swedish chemist Johann Gottlieb Gahn, who extracted it from the mineral pyrolusite. Still, the name Manganum does not derive from Gahn or the mineral itself. But instead, it traces back to the Swedish province of Mánland (now part of modern-day Sweden), where the mineral was initially discovered. The term Mánland was later Latinized to Manganum, and this became the accepted name for the element Turns out it matters..
The choice of Mn as the symbol is a direct reflection of this Latin origin. In the early 19th century, chemists began standardizing elemental symbols, often using the first letter of the element’s name in Latin. For manganese, the first letter of Manganum is M, but since another element, magnesium, also starts with M, a unique identifier was needed. The second letter, n, was chosen to distinguish it, resulting in the symbol Mn. This practice of using Latin names to derive symbols is common in chemistry, as seen with elements like Fe (from Ferrum for iron) or Au (from Aurum for gold).
Chemical Properties and the Role of Manganese
To fully grasp the significance of the symbol Mn, Understand the element itself — this one isn't optional. Manganese (Mn) is a transition metal with an atomic number of 25, meaning it has 25 protons in its nucleus. Here's the thing — it is located in Group 7 of the periodic table and is known for its ability to exhibit multiple oxidation states, ranging from +2 to +7. This versatility makes manganese a critical component in various chemical reactions and industrial applications It's one of those things that adds up. Still holds up..
One of manganese’s most notable properties is its role in forming alloys. When combined with iron, it creates manganese steel, which is highly resistant to wear and corrosion. But this property has made manganese a staple in the production of tools, machinery, and even surgical instruments. Additionally, manganese is a key element in the manufacturing of batteries, particularly in lithium-ion and alkaline batteries, where manganese dioxide (MnO₂) serves as a cathode material It's one of those things that adds up..
Worth pausing on this one.
Beyond industrial uses, manganese plays a vital role in biological systems. Which means it is an essential trace element for humans and other organisms, functioning as a cofactor in enzymes involved in metabolic processes. Plus, for example, the enzyme manganese superoxide dismutase (MnSOD) helps neutralize harmful free radicals in the body, protecting cells from oxidative damage. Without manganese, many biological processes would be impaired, highlighting its importance in both health and environmental contexts.
Worth pausing on this one.
The Symbol Mn in Chemical Communication
The symbol Mn is more than just a label; it is a universal shorthand that facilitates clear communication in chemistry. Even so, in chemical formulas, Mn denotes the presence of manganese atoms in compounds. Take this: in the compound manganese dioxide (MnO₂), the Mn symbol clearly identifies the element involved. This standardization is crucial for scientists, educators, and students to avoid confusion and ensure accuracy in research and teaching And that's really what it comes down to..
The International Union of Pure and Applied Chemistry (IUPAC) oversees the standardization of elemental symbols, ensuring consistency across the globe. So naturally, this uniformity is vital in a field where precise terminology is necessary for reproducibility and collaboration. The symbol Mn adheres to IUPAC guidelines, which dictate that symbols should be derived from the element’s name in its original language, often Latin. This approach not only honors the historical roots of chemical nomenclature but also provides a logical framework for naming new elements as they are discovered Simple as that..
Manganese in Everyday Life: From Soil to Technology
The symbol Mn might seem abstract, but its implications are deeply embedded in everyday life. Manganese is present in soil, water, and even the
human body, where it supports bone formation and helps metabolize nutrients like vitamin K and cholesterol. Dietary sources such as nuts, whole grains, and leafy greens provide manganese to the diet, though excessive intake can be toxic, underscoring the delicate balance required for optimal health Worth keeping that in mind. Took long enough..
In technology, manganese’s magnetic properties make it indispensable in the production of ferrite cores used in transformers and computer hard drives. Day to day, meanwhile, in the environment, manganese is released through weathering of rocks and mining activities, playing a role in soil fertility for plants. Worth adding: its ability to retain magnetic fields while resisting corrosion ensures efficiency in electronic components. Still, improper disposal of manganese-containing waste can lead to pollution, highlighting the need for sustainable practices in its extraction and use Took long enough..
As modern society advances, manganese’s versatility continues to shape industries, from renewable energy systems to medical devices. On top of that, its dual role as a biological essential and industrial workhorse underscores its enduring significance. By bridging the microscopic world of enzymes and the macro scale of global manufacturing, Mn stands as a testament to the interconnectedness of science and daily life.
To wrap this up, manganese is far more than a mere element on the periodic table. Its unique properties, from alloy strength to enzymatic function, make it a cornerstone of both natural processes and human innovation. As we work through challenges in health, technology, and sustainability, the story of manganese reminds us that even the smallest elements can have the largest impact Most people skip this — try not to..
Quick note before moving on.
Future Horizons: Manganese in Emerging Technologies
As the world pivots toward sustainable solutions, manganese is poised to play an increasingly critical role. Which means in the realm of energy storage, researchers are exploring manganese-based batteries as a cost-effective and environmentally friendly alternative to traditional lithium-ion systems. These batteries make use of manganese’s abundance and lower toxicity, offering potential breakthroughs in electric vehicles and grid-scale energy storage. Additionally, manganese oxide compounds are being investigated for their catalytic properties in hydrogen production, a critical component of the emerging hydrogen economy.
Worth pausing on this one.
In medicine, manganese’s magnetic properties are revolutionizing diagnostic imaging. Manganese-enhanced magnetic resonance imaging (MEMRI) allows for real-time tracking of neural activity and tumor progression, providing clinicians with unprecedented precision. Meanwhile, advancements in biotechnology are uncovering manganese’s potential in targeted drug delivery systems, where its ions can be engineered to bind specific cellular receptors, enhancing treatment efficacy while minimizing side effects.
Environmental remediation also stands to benefit from manganese’s unique chemistry. Scientists are developing manganese oxide nanoparticles capable of breaking down pollutants in water and soil, offering a promising tool
Manganese’s adaptability extends beyond its natural and industrial roles, as its applications in up-to-date technologies continue to evolve. But in renewable energy, the element is being harnessed to improve the efficiency of solar panels and fuel cells, reinforcing its value in combating climate change. Its ability to enhance the conductivity of certain materials also makes it a key player in next-generation electronics, where performance and sustainability are key.
Understanding these advancements underscores a broader narrative: manganese is not just a chemical component but a catalyst for innovation. From powering cleaner transportation to refining medical diagnostics, its influence permeates various sectors, shaping a future where science and necessity align.
The short version: the journey of manganese from earth to innovation reflects its profound significance. As research progresses, its potential will only deepen, offering solutions that balance human progress with ecological responsibility.
This evolving story emphasizes the importance of continuing to explore and appreciate the elements that underpin modern advancements.
Conclusion: Manganese exemplifies the involved link between nature and technology, reminding us that even the smallest elements can drive transformative change. Its continued study and responsible use will be essential in building a sustainable and innovative world That's the part that actually makes a difference..