What Is The Molar Mass Of Nitrogen Gas
tweenangels
Mar 14, 2026 · 8 min read
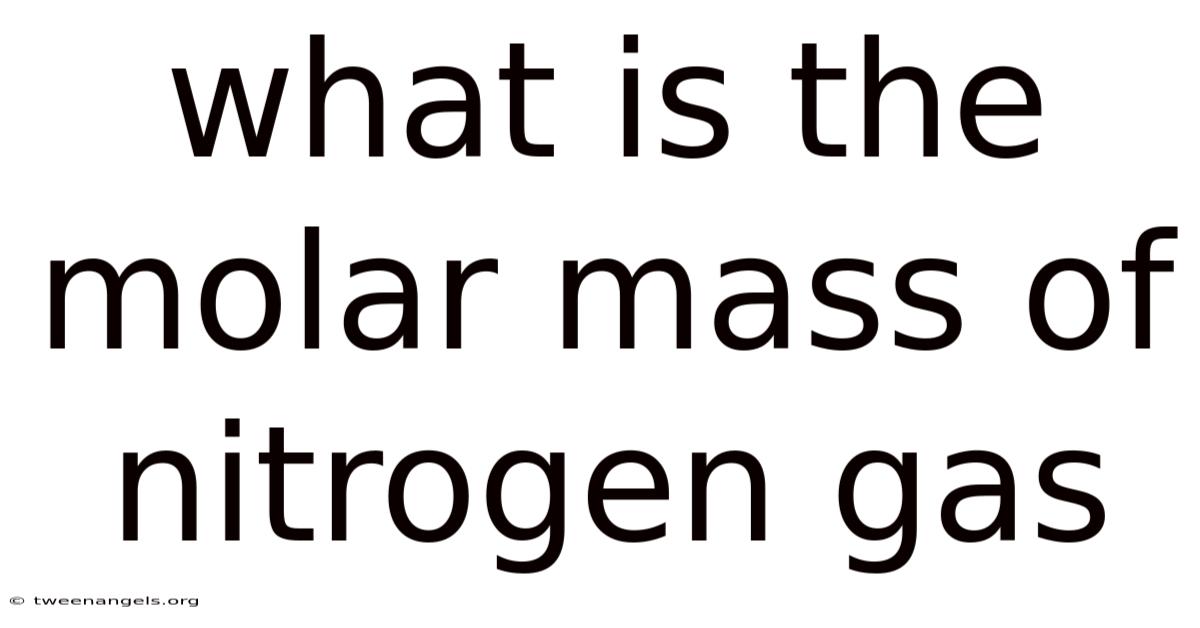
Table of Contents
What Is the Molar Mass of Nitrogen Gas?
The molar mass of nitrogen gas (N₂) is a fundamental concept in chemistry that plays a critical role in understanding chemical reactions, gas behavior, and stoichiometric calculations. Nitrogen gas, which makes up approximately 78% of Earth’s atmosphere, exists as diatomic molecules (N₂) under standard conditions. Its molar mass, defined as the mass of one mole of a substance, is essential for converting between the mass of a substance and the number of moles—a key step in many scientific and industrial applications.
This article explores the molar mass of nitrogen gas, how it is calculated, and its significance in chemistry and beyond.
Understanding Molar Mass
Before diving into nitrogen gas specifically, it’s important to grasp the broader concept of molar mass. Molar mass is the mass of one mole of a pure substance, expressed in grams per mole (g/mol). For elements, the molar mass is numerically equal to the atomic mass listed on the periodic table. However, for compounds or molecules like nitrogen gas (N₂), the molar mass is calculated by summing the atomic masses of all atoms in the molecule.
For example, nitrogen gas consists of two nitrogen atoms bonded together. Since each nitrogen atom has an atomic mass of approximately 14.01 atomic mass units (amu), the molar mass of N₂ is twice that value.
Calculating the Molar Mass of Nitrogen Gas
To determine the molar mass of nitrogen gas, follow these steps:
-
Identify the atomic mass of nitrogen:
From the periodic table, the atomic mass of nitrogen (N) is 14.01 g/mol. This value represents the average mass of all nitrogen isotopes, weighted by their natural abundance. -
Account for the diatomic nature of nitrogen gas:
Nitrogen gas exists as N₂ molecules, meaning each molecule contains two nitrogen atoms. To calculate the molar mass of N₂, multiply the atomic mass of nitrogen by 2:
$ \text{Molar mass of N₂} = 2 \times 14.01 , \text{g/mol} = 28.02 , \text{g/mol} $ -
Verify the result:
Double-check the calculation using reliable sources or periodic tables to ensure accuracy. The molar mass of nitrogen gas is consistently reported as 28.02 g/mol in scientific literature.
This value is crucial for stoichiometric calculations, such as determining how much nitrogen gas is produced or consumed in a chemical reaction.
Why Is the Molar Mass of Nitrogen Gas Important?
The molar mass of nitrogen gas has wide-ranging applications in chemistry, biology, and environmental science. Here are a few key reasons why it matters:
-
Stoichiometry in Chemical Reactions:
In chemical equations, molar mass allows scientists to convert between the mass of reactants and products and the number of moles. For instance, in the Haber process (used to synthesize ammonia), knowing the molar mass of N₂ helps calculate the exact amounts of nitrogen and hydrogen gas needed. -
Gas Laws and Physical Properties:
The molar mass of a gas influences its density, diffusion rate, and behavior under pressure. For example, the density of nitrogen gas can be calculated using the formula:
$ \text{Density} = \frac{\text{Molar mass}}{\text{Molar volume}} $
At standard temperature and pressure (STP), one mole of any gas occupies 22.4 liters. Thus, the density of N₂ at STP is:
$ \text{Density} = \frac{28.02 , \text{g/mol}}{22.4 , \text{L/mol}} \approx 1.25 , \text{g/L} $ -
Environmental and Industrial Relevance:
Nitrogen gas is a major component of the Earth’s atmosphere. Its
Its abundance makes it a key player in the global nitrogen cycle, where atmospheric N₂ is fixed into biologically usable forms by nitrogen‑fixing microorganisms and, to a lesser extent, by lightning. This fixed nitrogen eventually returns to the atmosphere through denitrification, completing a loop that regulates soil fertility, plant productivity, and the composition of aquatic ecosystems.
In industry, the inert nature of nitrogen gas is exploited to create oxygen‑free environments that prevent oxidation or combustion. It is used as a blanketing gas in the storage of reactive chemicals, as a purge medium in semiconductor fabrication, and as a carrier gas in chromatographic analyses. Food‑packaging operations rely on nitrogen to displace oxygen, thereby extending shelf life and preserving flavor. Cryogenic applications take advantage of nitrogen’s low boiling point (‑196 °C) to achieve rapid freezing of biological samples and to supercool superconducting magnets.
From an environmental perspective, accurate knowledge of N₂’s molar mass enables precise quantification of nitrogen fluxes in atmospheric models. When estimating emissions of nitrogen oxides (NOₓ) or nitrous oxide (N₂O) from combustion processes, scientists first convert measured masses to moles using the 28.02 g mol⁻¹ value, then apply stoichiometric relationships to trace the fate of nitrogen species. This conversion is essential for assessing air‑quality impacts, greenhouse‑gas inventories, and the effectiveness of mitigation strategies such as selective catalytic reduction in power plants.
In summary, the molar mass of nitrogen gas — 28.02 g mol⁻¹ — serves as a fundamental constant that bridges microscopic atomic properties with macroscopic chemical behavior. Its correct application underpins stoichiometric calculations, gas‑law predictions, industrial process design, and environmental monitoring. By mastering this simple yet powerful datum, chemists, engineers, and scientists can reliably predict reaction outcomes, optimize material handling, and quantify the role of nitrogen in both natural and engineered systems.
Building onthis foundation, researchers are now leveraging the precise 28.02 g mol⁻¹ conversion to explore more sophisticated applications. In isotopic labeling studies, for instance, the same molar‑mass constant allows scientists to track ^15N incorporation into biomolecules with sub‑percent accuracy, revealing metabolic pathways that were previously obscured. Such data are indispensable for designing bio‑engineered pathways that convert atmospheric N₂ into value‑added chemicals, a strategy that could dramatically reduce the energy intensity of fertilizer production.
The same stoichiometric precision also underpins emerging technologies in nitrogen‑based energy storage. Liquid organic nitrogen carriers, such as ammonia‑borane derivatives, are being evaluated as hydrogen‑rich fuels for fuel‑cell systems. By converting measured quantities of these carriers into moles using the 28.02 g mol⁻¹ reference, engineers can predict heat of reaction, vapor pressure curves, and decomposition kinetics with confidence, accelerating the scale‑up of carbon‑neutral propulsion concepts.
Beyond the laboratory, the molar‑mass constant is reshaping how we monitor atmospheric nitrogen cycles in real time. Satellite spectrometers now retrieve column‑integrated N₂ concentrations by inverting spectral absorption features that depend on the exact molecular weight of N₂. When these retrievals are calibrated against ground‑based flask measurements anchored to 28.02 g mol⁻¹, the resulting datasets achieve the consistency needed to validate climate models that predict how rising temperatures will alter nitrogen fixation rates and, consequently, ecosystem carbon balances.
Looking ahead, the integration of high‑precision molar‑mass calculations into artificial‑intelligence‑driven reaction‑network simulations promises to accelerate discovery across chemistry, materials science, and environmental engineering. By feeding accurate 28.02 g mol⁻¹‑based stoichiometric constraints into predictive algorithms, we can anticipate side‑product formation, optimize catalyst design, and even forecast the long‑term fate of nitrogen emissions in a changing climate.
In closing, the seemingly simple figure of 28.02 g mol⁻¹ does far more than quantify a molecule; it serves as a universal conversion key that unlocks quantitative insight across scales — from the atom to the globe. Mastery of this constant empowers scientists and engineers to translate theoretical principles into practical solutions, ensuring that nitrogen’s abundant presence continues to drive innovation while safeguarding the planet’s delicate chemical equilibria.
Continuing theexploration of nitrogen's fundamental role, the precision afforded by the molar-mass constant extends into the realm of quantum chemistry and materials design. High-fidelity calculations of reaction energetics and transition states, critical for designing novel catalysts or nitrogen-rich materials with tailored electronic properties, rely fundamentally on accurate molecular weights. The 28.02 g/mol figure provides the essential bridge between the abstract quantum mechanical models describing electron interactions and the tangible, measurable quantities required for synthesis and characterization. This precision enables the prediction of novel phases, the optimization of nitrogen fixation catalysts beyond current enzymatic or industrial limits, and the design of materials for advanced applications like high-capacity hydrogen storage or nitrogen-based semiconductors.
Furthermore, the constant underpins critical environmental monitoring and policy frameworks. As satellite-based remote sensing techniques become increasingly sophisticated in detecting trace atmospheric constituents like nitrous oxide (N₂O) – a potent greenhouse gas and ozone-depleting agent – the absolute accuracy of the N₂ molecular weight (28.02 g/mol) remains the bedrock for converting spectral data into reliable concentration measurements. These measurements, when integrated into global atmospheric models, provide the empirical foundation for assessing the effectiveness of mitigation strategies, such as optimized fertilizer application practices or advanced wastewater treatment technologies, aimed at curbing anthropogenic nitrogen emissions and their contribution to climate change and ecosystem disruption.
In essence, the molar-mass constant of nitrogen transcends its simple numerical value. It is the indispensable quantitative language that allows disparate scientific disciplines – from the quantum scale of atomic interactions to the planetary scale of climate systems – to communicate, collaborate, and innovate. Mastery of this constant empowers researchers to translate theoretical understanding into tangible technologies, driving progress in sustainable agriculture, clean energy, and environmental protection. By ensuring the accuracy of stoichiometric relationships and molecular quantification, the 28.02 g/mol figure safeguards the integrity of scientific discovery and engineering solutions, enabling humanity to harness nitrogen's abundance responsibly while preserving the delicate chemical equilibria that sustain life on Earth.
Conclusion: The molar-mass constant of nitrogen (28.02 g/mol) is far more than a mere conversion factor; it is the foundational quantitative keystone underpinning progress across the entire spectrum of scientific and technological endeavor. From tracking metabolic pathways in biochemistry to enabling carbon-neutral propulsion, from calibrating climate models to designing next-generation materials, its precision unlocks quantitative insight and drives innovation. By providing the essential link between molecular theory and measurable reality, this constant empowers scientists and engineers to translate fundamental principles into practical solutions that address global challenges in sustainability, energy, and environmental stewardship, ensuring nitrogen's vital role continues to foster progress while safeguarding the planet's future.
Latest Posts
Latest Posts
-
Human Biology Concepts And Current Issues
Mar 14, 2026
-
Fundamentals Of Futures And Options Markets
Mar 14, 2026
-
What Observable Cellular Process Explains Mendels Law Of Segregation
Mar 14, 2026
-
The Vertebrate Lung First Appeared In
Mar 14, 2026
-
Which Of The Following Is An Example Of Positive Feedback
Mar 14, 2026
Related Post
Thank you for visiting our website which covers about What Is The Molar Mass Of Nitrogen Gas . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.