What Is The Molar Mass Of C12h22o11
tweenangels
Mar 14, 2026 · 6 min read
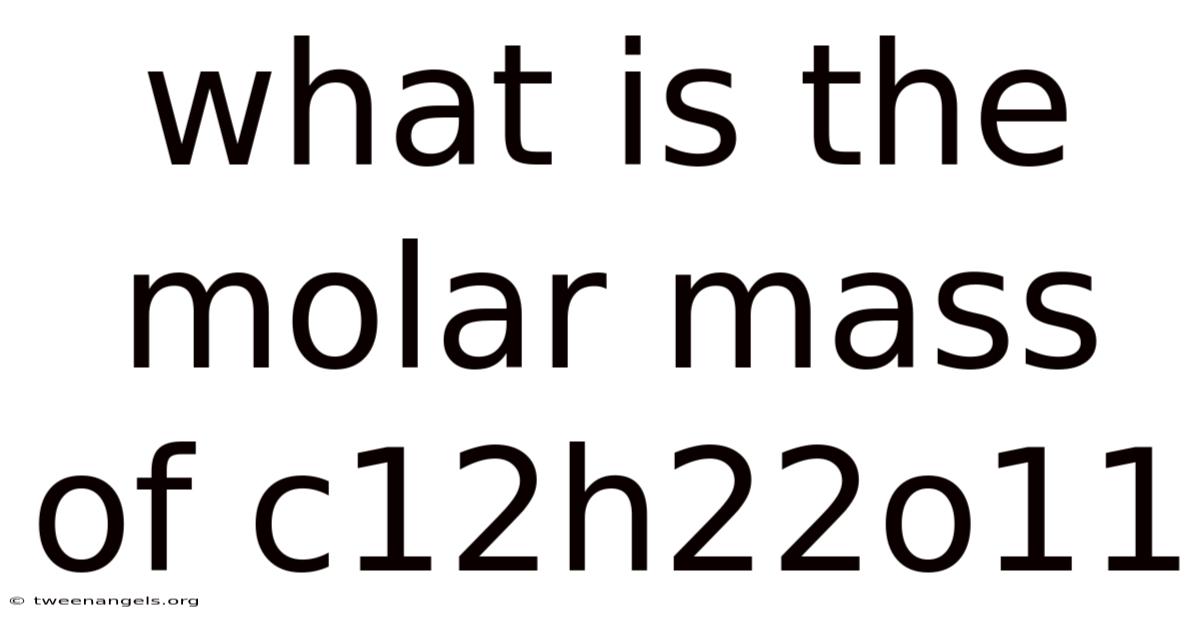
Table of Contents
Understanding the Molar Mass of C₁₂H₂₂O₁₁ (Sucrose)
The molar mass of a compound is a fundamental concept in chemistry, bridging the gap between the microscopic world of atoms and molecules and the measurable quantities we use in the laboratory. For the molecule with the chemical formula C₁₂H₂₂O₁₁, commonly known as sucrose or table sugar, determining its molar mass is a precise exercise in summing the contributions of its constituent atoms. The calculated molar mass of sucrose is 342.30 g/mol (grams per mole). This single value is a cornerstone for countless calculations, from preparing solutions in a high school lab to designing industrial fermentation processes. This article will deconstruct the calculation, explore the scientific principles behind it, and illuminate why this number is so critically important across scientific and industrial fields.
What is Molar Mass? The Bridge to the Macro World
Before calculating, it is essential to understand what molar mass represents. Molar mass is the mass of one mole of a given substance. A mole, in turn, is the SI base unit for amount of substance, defined as containing exactly 6.02214076×10²³ elementary entities (atoms, molecules, ions, etc.). This number is known as Avogadro's constant.
Therefore, the molar mass of a compound tells us the mass in grams of 6.022×10²³ molecules of that compound. For an element, it is numerically equal to its atomic mass in atomic mass units (amu) but expressed in grams per mole. For a compound, it is the sum of the molar masses of all atoms in its chemical formula. This conversion factor allows chemists to weigh out precise numbers of molecules, making quantitative chemistry possible.
Step-by-Step Calculation for C₁₂H₂₂O₁₁
The formula C₁₂H₂₂O₁₁ reveals the composition of a single sucrose molecule: 12 carbon atoms, 22 hydrogen atoms, and 11 oxygen atoms. To find the total molar mass, we must sum the molar masses of these individual atoms.
We use the standard atomic weights from the periodic table, which reflect the weighted average mass of an element's naturally occurring isotopes.
- Carbon (C): 12.011 g/mol (often rounded to 12.01 g/mol for general calculations)
- Hydrogen (H): 1.008 g/mol
- Oxygen (O): 15.999 g/mol (often rounded to 16.00 g/mol)
The calculation proceeds as follows:
- Mass contribution from Carbon: 12 atoms × 12.011 g/mol = 144.132 g/mol
- Mass contribution from Hydrogen: 22 atoms × 1.008 g/mol = 22.176 g/mol
- Mass contribution from Oxygen: 11 atoms × 15.999 g/mol = 175.989 g/mol
Total Molar Mass = (12 × 12.011) + (22 × 1.008) + (11 × 15.999) Total Molar Mass = 144.132 + 22.176 + 175.989 Total Molar Mass = 342.297 g/mol
Rounding to two decimal places, the standard molar mass of sucrose is 342.30 g/mol. It is crucial to use consistent significant figures based on the precision of the atomic weights used. The slight variation (342.297 vs. 342.30) depends on the specific atomic mass values sourced from the periodic table.
The Scientific Significance of Sucrose's Molar Mass
This number, 342.30 g/mol, is not just an abstract figure. It is a key that unlocks practical applications.
- Stoichiometry and Chemical Reactions: In any reaction involving sucrose, such as its hydrolysis into glucose and fructose (C₁₂H₂₂O₁₁ + H₂O → C₆H₁₂O₆ + C₆H₁₂O₆), the molar mass allows for the conversion between mass and moles. If a recipe calls for 2 moles of sucrose, you would precisely weigh out 2 mol × 342.30 g/mol = 684.60 grams. Conversely, if you start with 100 grams of sucrose, you can calculate that you have 100 g / 342.30 g/mol ≈ 0.292 moles available for reaction.
- Solution Preparation: Molarity (M), a core concentration unit, is defined as moles of solute per liter of solution. To make 1 liter of a 0.5 M sucrose solution, you would dissolve 0.5 mol × 342.30 g/mol = 171.15 grams of pure sucrose in water and then dilute to the 1-liter mark. The molar mass is the indispensable conversion factor here.
- Industrial and Biological Contexts: Sucrose is a primary feedstock in biofuel production (fermentation to ethanol), food science, and biochemistry. Engineers and scientists constantly convert between mass (tons of sugar processed) and moles (to calculate theoretical yields of ethanol or other metabolites) using this molar mass. In cellular biology, understanding the molar concentration of sugars in the bloodstream or in a culture medium relies on this fundamental property.
Common Misconceptions and Pitfalls
When calculating or using the molar mass of C₁₂H₂₂O₁₁, several errors frequently occur.
- Confusing Molar Mass with Molecular Mass/Formula Mass: The molecular mass (or formula mass) is the sum of the atomic masses in atomic mass units (amu) for a single molecule. For sucrose, this is also 342.30 amu. The molar mass is the mass of one mole of those molecules, expressed in grams per mole (g/mol). Numerically, they are identical, but the units and scale are fundamentally different.
- Using Incorrect Atomic Masses: Relying on rounded atomic masses (e.g., C=12, H=1, O=16) yields a molar mass of 342 g/mol. While this is a useful estimate, precise scientific work requires the more accurate values (12.011, 1.008, 15.999) to avoid cumulative error, especially in large-scale or sensitive analytical contexts.
Beyond these applications and pitfalls, sucrose's molar mass plays a critical role in understanding colligative properties—those that depend on the number of solute particles, not their identity. For instance, when calculating the boiling point elevation or freezing point depression of a sugar solution, the essential input is the molality (moles of solute per kg of solvent), which is derived directly from the molar mass. A precise 342.30 g/mol value ensures accurate predictions for processes like candy making (where sugar concentration controls texture and crystallization) or cryopreservation.
Furthermore, in analytical chemistry, the molar mass is indispensable for determining purity. If a sample labeled as "sucrose" has a measured melting point significantly lower than the pure compound's 186°C, one can use the observed depression and the known i factor (van't Hoff factor, which is 1 for sucrose) along with the molar mass to estimate the molar concentration of impurities. This application turns the molar mass into a diagnostic tool for quality control in food and pharmaceutical industries.
Educationally, the calculation and consistent use of sucrose's molar mass serve as a foundational exercise for students, reinforcing the mole concept, dimensional analysis, and the importance of significant figures. It transforms the abstract periodic table into a practical instrument for quantitative reasoning.
Conclusion
Thus, the molar mass of sucrose, 342.30 g/mol, is far more than a static number on a data sheet. It is a dynamic and essential conversion factor that underpins quantitative work across the chemical sciences and engineering. From the precise formulation of a laboratory buffer to the industrial-scale fermentation of biofuels, from predicting the physical behavior of solutions to verifying the purity of a commodity, this value acts as a fundamental bridge between the measurable mass of a substance and the invisible world of molecules and moles. Its accurate determination and application remain a cornerstone of both theoretical understanding and practical innovation in chemistry.
Latest Posts
Latest Posts
-
What Type Of Biological Molecule Is Dna Helicase
Mar 14, 2026
-
Pdms 3 Online Scoring And Report System
Mar 14, 2026
-
Pearson Support Phone Number 24 7
Mar 14, 2026
-
How Many Valence Electrons Does Se Have
Mar 14, 2026
-
Which Of The Following Hormones Has Intracellular Receptors
Mar 14, 2026
Related Post
Thank you for visiting our website which covers about What Is The Molar Mass Of C12h22o11 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.