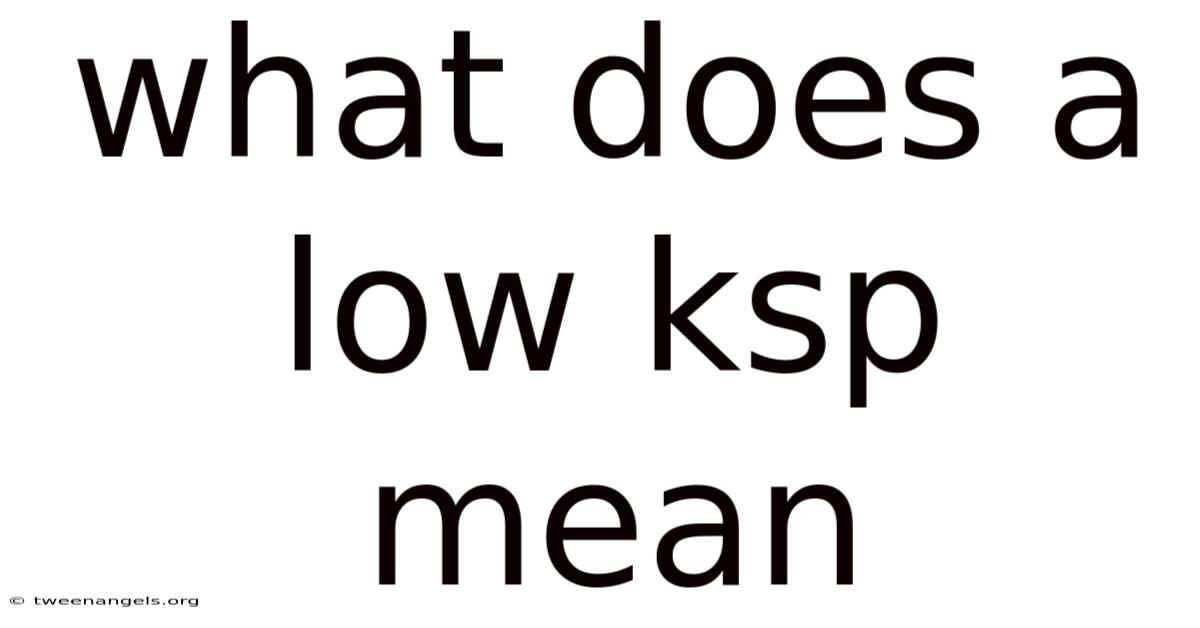Alow Ksp value signals that a sparingly soluble salt dissolves only minimally in water, meaning that the equilibrium between the solid and its ions lies far to the left; in other words, what does a low ksp mean is that the compound is barely soluble.
Understanding solubility product constants (Ksp) is essential for anyone studying chemistry, geology, environmental science, or even pharmaceuticals. Still, while the symbol itself may appear in textbooks as a simple number, its implications ripple through countless real‑world applications. This article unpacks the concept of a low Ksp, explains why it matters, and provides practical examples that illustrate how chemists interpret and exploit these tiny equilibrium constants.
What Is Ksp and Why Does It Matter?
The solubility product constant, denoted Ksp, quantifies the maximum amount of a sparingly soluble ionic compound that can dissolve in water at a given temperature. It is derived from the concentrations of the constituent ions when the dissolution process reaches equilibrium:
Worth pausing on this one.
[ \text{AB(s)} \rightleftharpoons \text{A}^+(aq) + \text{B}^-(aq) \quad\Rightarrow\quad K_{sp}= [\text{A}^+][\text{B}^-] ]
Because Ksp is a product of ion concentrations, it is unit‑less and typically expressed in scientific notation (e.g.Now, , (1. 8 \times 10^{-10})). A high Ksp (on the order of (10^{-2}) to (10^{0})) indicates that a salt dissolves readily, whereas a low Ksp (often (10^{-10}) or smaller) points to a compound that remains largely undissolved Worth keeping that in mind..
Interpreting a Low Ksp Value
1. Quantitative Meaning
When chemists ask what does a low ksp mean, they are usually seeking a concrete interpretation:
- Low Ksp ≈ Low solubility – Only a minuscule fraction of the solid will dissolve before the solution becomes saturated.
- Magnitude matters – A Ksp of (10^{-12}) is far less soluble than one of (10^{-5}), even though both are “low” compared to highly soluble salts like NaCl (Ksp ≈ (36)).
- Temperature dependence – Ksp values are temperature‑specific; a low Ksp at 25 °C may increase at higher temperatures, altering the solubility behavior.
2. Qualitative Implications
- Saturated solutions are rare – If you attempt to dissolve a salt with a very low Ksp, the solution will become saturated after only a trace amount of ions is released.
- Precipitation propensity – Low Ksp compounds are prone to precipitate when ions are brought together, a principle exploited in qualitative analysis and environmental remediation.
- Stability of solids – In geological contexts, minerals with low Ksp can persist for eons because they dissolve only infinitesimally under natural conditions.
Factors That Influence Ksp Values
1. Chemical Nature of the Salt
- Charge magnitude – Ions with higher absolute charges tend to attract each other more strongly, resulting in lower Ksp values. Here's one way to look at it: MgO (Mg²⁺ and O²⁻) has a much lower Ksp than NaCl (Na⁺ and Cl⁻).
- Lattice energy – The strength of the ionic lattice directly impacts solubility; higher lattice energy translates to a lower Ksp.
2. Solvent Characteristics
- Temperature – Raising temperature generally increases Ksp for most salts, though exceptions exist.
- Presence of complexing agents – Ligands that bind to one of the ions can effectively lower the ion concentration, shifting equilibrium and altering the apparent Ksp.
3. Ionic Strength of the Solution
- Adding other electrolytes changes the ionic strength, which can affect activity coefficients and thus the observed Ksp. This is especially relevant in high‑concentration environments like seawater.
Practical Examples of Low Ksp Compounds
| Compound | Ksp (≈) | Typical Use / Occurrence |
|---|---|---|
| AgCl | (1.Still, 8 \times 10^{-10}) | Photographic film, silver plating |
| PbSO₄ | (1. 6 \times 10^{-8}) | Battery acid, lead‑acid batteries |
| CaCO₃ | (3. |
These examples demonstrate how low Ksp values manifest across different domains, from industrial processes to natural mineral formation It's one of those things that adds up..
How Chemists Use Low Ksp Information
1. Predicting Precipitation
When mixing solutions containing ions that could form an insoluble product, the ion product (Q) is calculated and compared to Ksp:
- If Q > Ksp, precipitation occurs.
- If Q < Ksp, the solution remains unsaturated.
This principle underlies qualitative analysis schemes and the design of selective precipitation reactions.
2. Designing Solubility Equilibria
In pharmaceuticals, controlling the dissolution rate of poorly soluble drugs is crucial. Formulating a drug as a salt with a higher Ksp (or using techniques like solid dispersions) can enhance bioavailability.
3. Environmental Modeling
Geologists model the dissolution of minerals like gypsum (CaSO₄·2H₂O) or calcite (CaCO₃) in groundwater. Low Ksp values dictate how quickly these minerals can supply ions to the water, influencing soil chemistry and karst formation.
Common Misconceptions About Low Ksp
- “Low Ksp means the compound is completely insoluble.”
Reality: Even with a Ksp of (10^{-30}), a tiny amount