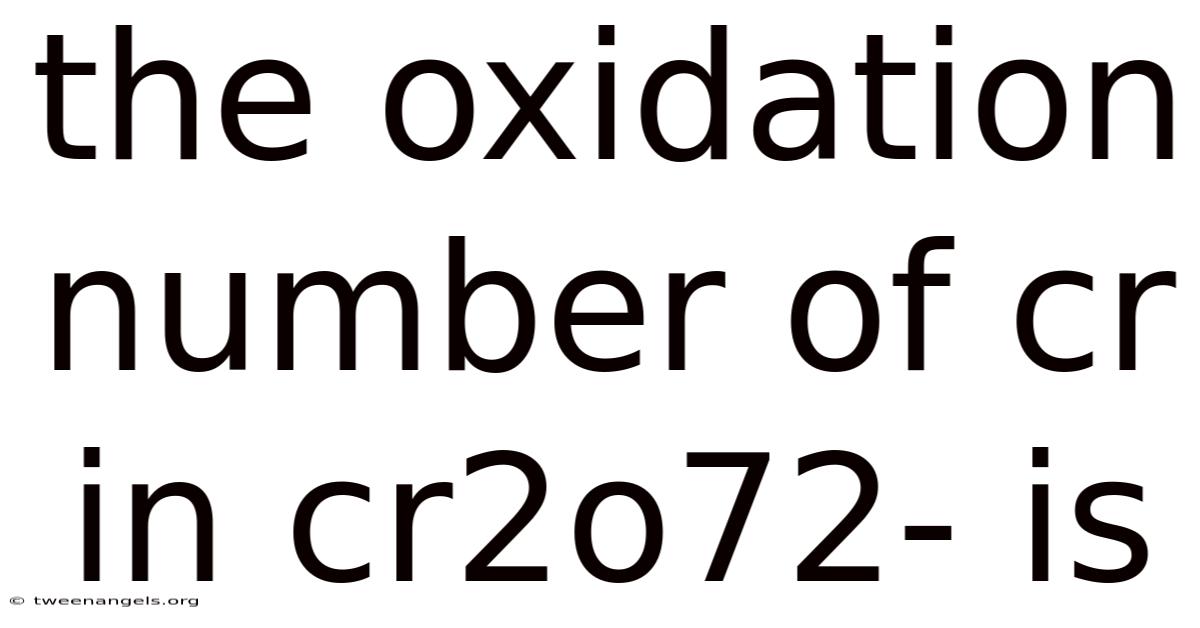The oxidationnumber of Cr in the dichromate ion Cr₂O₇²⁻ is +6, and determining this value requires a systematic application of oxidation‑state rules that illuminate the redox behavior of chromium in acidic solutions. This article walks you through the logical steps, explains the underlying science, and answers common questions, giving you a complete picture of why chromium carries a +6 charge in Cr₂O₇²⁻ Less friction, more output..
And yeah — that's actually more nuanced than it sounds.
Introduction
Understanding the oxidation number of Cr in Cr₂O₇²⁻ is fundamental for anyone studying inorganic chemistry, redox reactions, or analytical methods. Also, the dichromate ion appears in countless laboratory procedures, from titrations to wastewater treatment, yet its charge originates from a precise balance of electron distribution among its atoms. By dissecting the rules that govern oxidation states, you can predict the charge on chromium without relying on memorization, gaining insight into the ion’s reactivity and its role in oxidation‑reduction processes Most people skip this — try not to..
Worth pausing on this one.
Steps to Determine the Oxidation Number Below is a concise, numbered procedure that you can follow each time you encounter a polyatomic ion like dichromate.
- Assign known oxidation numbers – Oxygen almost always has an oxidation number of –2, while hydrogen is +1.
- Set up an algebraic equation – The sum of all oxidation numbers in the ion must equal the overall charge of the species.
- Solve for the unknown oxidation number – In Cr₂O₇²⁻, let the oxidation number of chromium be x; then 2x + 7*(–2) = –2.
- Calculate – 2x – 14 = –2 → 2x = 12 → x = +6.
- Verify consistency – Check that the calculated value satisfies all rules, especially the requirement that the total charge matches the ion’s known charge. Key takeaway: The algebraic approach transforms a seemingly complex problem into a straightforward calculation, reinforcing why the oxidation number of Cr in Cr₂O₇²⁻ is definitively +6.
Scientific Explanation
Why Oxidation Numbers Matter
Oxidation numbers are a bookkeeping tool that chemists use to track electron transfer in chemical reactions. They do not represent actual charges on individual atoms, but they provide a clear framework for predicting how substances behave in redox processes. In the case of dichromate, the +6 oxidation state of chromium makes it a powerful oxidizing agent, capable of accepting electrons from a wide range of reducing agents The details matter here..
Electronic Configuration and Stability
Chromium’s ground‑state electron configuration is [Ar] 3d⁵ 4s¹. Here's the thing — this loss yields a highly stable, compact ion that is energetically favored in acidic environments where the dichromate ion exists predominantly as Cr₂O₇²⁻. When it attains a +6 oxidation state, it loses all six valence electrons (the single 4s electron and five 3d electrons), resulting in a configuration that resembles the noble gas argon ([Ar]). The stability is further enhanced by the strong Cr–O covalent bonds formed with the seven oxygen atoms, each contributing to the overall charge distribution.
Role of Acidic Conditions
The oxidation state of chromium in Cr₂O₇²⁻ is most commonly observed in acidic solutions. Consider this: under basic conditions, dichromate converts to chromate (CrO₄²⁻), where each chromium atom has an oxidation number of +6 as well, but the overall geometry and bonding differ. The prevalence of the +6 state in both forms underscores its thermodynamic favorability across a range of pH values, though the speciation changes with pH.
Redox Implications
Because chromium is in the +6 state, it can be reduced to lower oxidation states such as +3 (forming Cr³⁺) or +2 (forming Cr²⁺). This reduction is the basis for many titrations, where dichromate serves as the oxidizing agent that accepts electrons from analytes like ferrous sulfate. The clear change in oxidation number provides a measurable endpoint, making the concept practically indispensable in analytical chemistry.
Frequently Asked Questions
What is the oxidation number of oxygen in Cr₂O₇²⁻?
Oxygen almost universally carries an oxidation number of –2 in most compounds, including the dichromate ion.
Can chromium have an oxidation number other than +6 in dichromate?
No. The algebraic sum of oxidation numbers must equal the ion’s overall charge, and the standard rules assign –2 to each oxygen, leading uniquely to +6 for chromium.
Why does the dichromate ion have a –2 charge?
The –2 charge arises from the combination of two chromium atoms each at +6 and seven oxygen atoms each at –2: 2(+6) + 7*(–2) = –2.*
How does the oxidation state of chromium affect the color of the dichromate ion?
The +6 oxidation state creates a charge‑transfer transition that imparts the characteristic orange color to Cr₂O₇²⁻ solutions.
Is the oxidation number of chromium the same in chromate (CrO₄²⁻)?
Yes. In chromate, each oxygen is still –2, giving 4(–2) = –8, so the chromium must be +6 to satisfy the overall –2 charge: +6 + (–8) = –2.*
Conclusion
The oxidation number of Cr in Cr₂O₇²⁻ is unequivocally +6, a result derived from
The oxidation number of Cr in Cr₂O₇²⁻ is unequivocally +6, a result derived from the formal charge balance that respects the universally accepted oxidation state of oxygen (–2) and the overall –2 charge of the dichromate ion. This +6 state is not merely a numerical artifact; it is a manifestation of the electronic structure that best accommodates the high oxidation potential of chromium while maintaining the geometry and ligand field of the polyatomic ion.
In practical terms, the +6 oxidation state endows dichromate with a powerful oxidizing capacity, which has made it a staple reagent in redox titrations, analytical chemistry, and industrial oxidation processes. Still, its stability in acidic media, coupled with the ease of reduction to lower oxidation states, allows chemists to manipulate electron flow with predictable precision. Conversely, the chromate ion (CrO₄²⁻) exemplifies how a shift in pH can alter speciation without changing the underlying oxidation state, underscoring the delicate interplay between thermodynamics and solution chemistry Surprisingly effective..
Understanding the oxidation state of chromium in dichromate is therefore essential not only for accurate stoichiometric calculations but also for anticipating the behavior of this ion in varied chemical environments—from laboratory titrations to large‑scale environmental remediation. The clarity of the +6 assignment serves as a foundation upon which chemists build more complex redox systems, design selective sensors, and develop greener oxidation protocols that balance efficacy with safety.
consistency with the established rules for assigning oxidation numbers. Each oxygen atom is assigned a –2 state, and with seven oxygens contributing a total of –14, the two chromium atoms must collectively balance this by summing to +12 to achieve the ion’s net –2 charge. As a result, each chromium is assigned an individual oxidation state of +6.
This formalism extends beyond a simple bookkeeping exercise; it reflects the underlying electronic distribution. The high electronegativity of oxygen stabilizes the negative charge, while the chromium centers operate in a high oxidation state that drives their reactivity. This reactivity is vividly demonstrated when dichromate is reduced, often transitioning to the +3 state, which shifts the solution color from orange to green—a visual cue familiar to countless chemists.
When all is said and done, the fixed +6 oxidation state in Cr₂O₇²⁻ is a cornerstone concept that reinforces the predictive power of oxidation number rules. It ensures consistency across chemical disciplines, from analytical procedures to industrial applications. By adhering to these principles, chemists can reliably interpret reaction mechanisms, balance complex redox equations, and design processes that take advantage of the potent oxidative properties of dichromate with controlled precision.
The official docs gloss over this. That's a mistake.