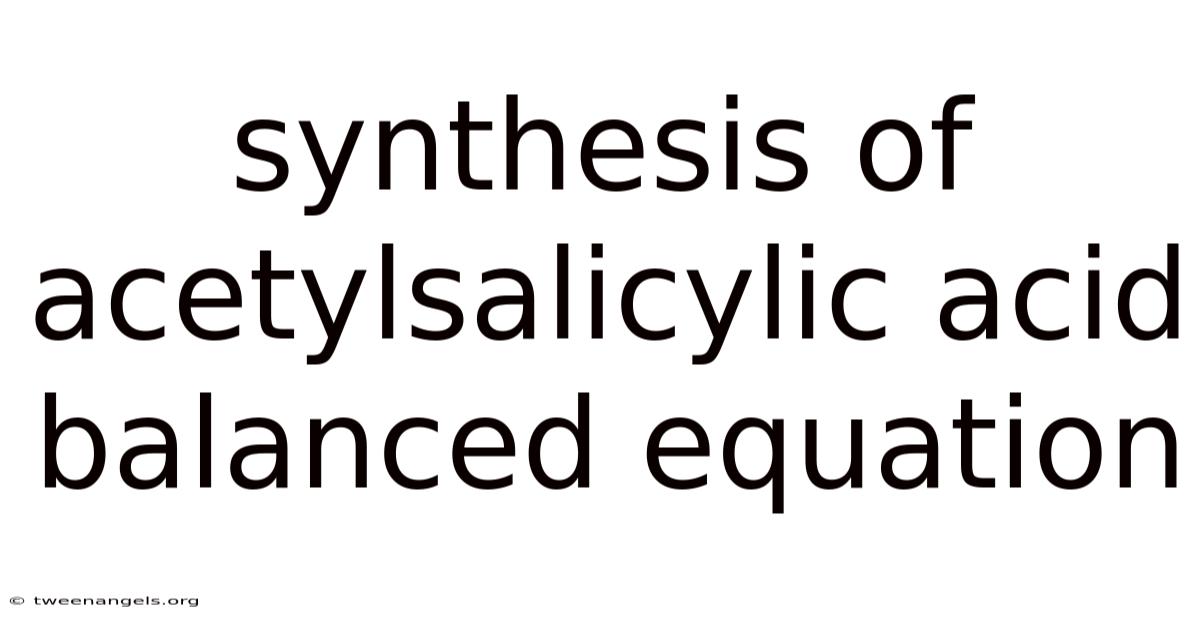Acetylsalicylic acid, better known as aspirin, is one of the most widely used medications in the world. And the synthesis of acetylsalicylic acid is a classic experiment often performed in organic chemistry laboratories, demonstrating the principles of esterification and acetylation. It is valued for its analgesic, anti-inflammatory, and antipyretic properties. Understanding the balanced chemical equation for this synthesis is essential for students, researchers, and anyone interested in pharmaceutical chemistry.
The process of synthesizing acetylsalicylic acid involves the reaction between salicylic acid and acetic anhydride. Salicylic acid is a phenolic compound with a hydroxyl group (-OH) attached to a benzene ring, while acetic anhydride is a reactive acylating agent. When these two compounds react, the hydroxyl group of salicylic acid is acetylated, resulting in the formation of acetylsalicylic acid (aspirin) and acetic acid as a byproduct.
The balanced chemical equation for the synthesis of acetylsalicylic acid is as follows:
C₇H₆O₃ (salicylic acid) + C₄H₆O₃ (acetic anhydride) → C₉H₈O₄ (acetylsalicylic acid) + C₂H₄O₂ (acetic acid)
This equation shows that one mole of salicylic acid reacts with one mole of acetic anhydride to produce one mole of acetylsalicylic acid and one mole of acetic acid. The reaction is typically carried out in the presence of a catalyst, such as concentrated sulfuric acid or phosphoric acid, which helps to speed up the reaction and increase the yield.
The mechanism of the reaction involves the nucleophilic attack of the hydroxyl group of salicylic acid on the carbonyl carbon of acetic anhydride. This results in the formation of a tetrahedral intermediate, which then collapses to release acetic acid and form the ester bond in acetylsalicylic acid. The reaction is an example of an acetylation reaction, where an acetyl group (CH₃CO-) is transferred from acetic anhydride to the hydroxyl group of salicylic acid.
In the laboratory, the synthesis of acetylsalicylic acid is often performed by mixing salicylic acid with acetic anhydride in a round-bottom flask. The mixture is then heated gently with stirring, and a few drops of concentrated sulfuric acid are added as a catalyst. In practice, after the reaction is complete, the mixture is poured into cold water to precipitate the acetylsalicylic acid. The product is then filtered, washed, and dried to obtain pure aspirin.
Counterintuitive, but true.
The balanced equation is not only important for understanding the stoichiometry of the reaction but also for calculating the theoretical yield and percent yield of the product. By knowing the molar masses of the reactants and products, one can determine how much aspirin should be produced from a given amount of salicylic acid and acetic anhydride. This information is crucial for optimizing the reaction conditions and improving the efficiency of the synthesis Not complicated — just consistent..
It is also worth noting that the synthesis of acetylsalicylic acid has some safety considerations. Which means, proper personal protective equipment, such as gloves, goggles, and a lab coat, should be worn when handling these chemicals. Acetic anhydride is a corrosive and lachrymatory substance, meaning it can cause severe irritation to the eyes and respiratory tract. Additionally, the reaction should be carried out in a well-ventilated area or under a fume hood to avoid inhalation of fumes Easy to understand, harder to ignore..
In a nutshell, the synthesis of acetylsalicylic acid is a fundamental experiment in organic chemistry that illustrates the principles of esterification and acetylation. The balanced chemical equation for this reaction is:
C₇H₆O₃ + C₄H₆O₃ → C₉H₈O₄ + C₂H₄O₂
This equation represents the transformation of salicylic acid and acetic anhydride into acetylsalicylic acid and acetic acid. Understanding this equation and the underlying chemistry is essential for students and researchers working in the field of pharmaceutical chemistry. By mastering the synthesis of acetylsalicylic acid, one gains valuable insights into the broader world of drug development and organic synthesis Not complicated — just consistent..
Isolation and Purification Techniques
After the crude aspirin has been filtered and dried, it is rarely pure enough for analytical or pharmaceutical use. Impurities such as unreacted salicylic acid, residual acetic anhydride, and side‑product acetic acid can remain embedded in the solid. Two common purification steps are employed:
-
Recrystallization – The crude product is dissolved in a minimum amount of hot ethanol or a 95 % ethanol–water mixture, then allowed to cool slowly to room temperature before being placed in an ice bath. As the solution cools, pure aspirin crystals nucleate and grow, while soluble impurities remain in the mother liquor. The crystals are collected by vacuum filtration, washed with a small amount of cold solvent, and dried. The choice of solvent system is guided by the principle that aspirin should be sparingly soluble at low temperature but readily soluble when hot Worth keeping that in mind..
-
Dry‑Ice/Acetone Wash – For a rapid “purity check,” the filtered solid can be briefly washed with a cold acetone‑dry‑ice slurry. Acetone removes surface‑adsorbed acetic acid and traces of acetic anhydride without dissolving the aspirin crystals, giving a whiter, more uniform product.
Both methods improve the percent purity, which can be quantified by melting‑point determination (pure aspirin melts at 135 °C ± 0.5 °C) or by thin‑layer chromatography (TLC) using a suitable solvent system (e.g.Consider this: , ethyl acetate : hexane 3 : 1). A single, sharp spot at the R_f value characteristic of aspirin confirms the absence of major contaminants.
Quantitative Analysis: Yield and Purity
To calculate the theoretical yield, one uses the limiting reagent—usually salicylic acid, because it is typically added in slight excess to drive the reaction to completion. To give you an idea, 5.00 g of salicylic acid (0.
[ \text{Theoretical mass of aspirin} = 0.Now, 0362\ \text{mol} \times 180. 16\ \frac{\text{g}}{\text{mol}} = 6.
If the isolated product after recrystallization weighs 5.80 g, the percent yield is:
[ % \text{Yield} = \frac{5.80\ \text{g}}{6.52\ \text{g}} \times 100 = 89% ]
Purity can be assessed by HPLC or GC‑MS. Which means a typical HPLC method uses a C18 reverse‑phase column with a gradient of water (0. 1 % formic acid) and acetonitrile. The area under the aspirin peak divided by the total area gives the purity percentage; values above 98 % are considered acceptable for most laboratory‑scale preparations.
Environmental and Waste‑Management Considerations
The synthesis generates several waste streams:
| Waste Stream | Primary Component | Recommended Disposal |
|---|---|---|
| Acetone wash solvent | Acetone | Collect in a labeled organic waste container; recycle if possible |
| Acidic aqueous filtrate | Acetic acid, residual sulfuric acid | Neutralize with sodium bicarbonate before disposal, then treat as aqueous waste |
| Solid residues (unreacted salicylic acid) | Salicylic acid | Can be recovered by recrystallization or disposed of as hazardous solid waste |
Minimizing waste is achievable by recycling the mother liquor from recrystallization—evaporating the solvent and reusing it for a second batch reduces both chemical consumption and cost.
Scale‑Up Considerations
When moving from a 5‑gram laboratory scale to a kilogram scale for pilot‑plant production, several parameters must be re‑examined:
- Heat Removal: The exothermic acetylation releases ~‑70 kJ mol⁻¹. On a larger scale, efficient cooling (e.g., jacketed reactors with controlled temperature ramps) is essential to avoid runaway reactions.
- Catalyst Loading: Sulfuric acid is effective at 0.5–1 % v/v in the lab, but on scale it can cause corrosion. Alternative solid acid catalysts (e.g., zeolites) or acid‑resistant reactors (stainless steel 316) are often employed.
- Process Control: In‑line IR or Raman spectroscopy can monitor the disappearance of the phenolic O–H stretch (~3300 cm⁻¹) and the emergence of the ester carbonyl (~1750 cm⁻¹), providing real‑time conversion data.
- Crystallization Engineering: Continuous crystallizers (e.g., mixed‑mode or MSMPR) give tighter control over crystal size distribution, which is critical for downstream tableting operations.
Pharmaceutical Relevance and Regulatory Perspective
Aspirin remains one of the most widely used non‑steroidal anti‑inflammatory drugs (NSAIDs). Modern formulations often demand high‑purity API (active pharmaceutical ingredient) meeting United States Pharmacopeia (USP) specifications: assay 98–102 % and impurity limits (e.g.But , salicylic acid ≤0. 1 %).
- Drying under vacuum to achieve moisture content <0.1 %,
- Polishing via recrystallization from aqueous ethanol, and
- Final sterilization (e.g., gamma irradiation) for sterile dosage forms.
Regulatory agencies also require a process validation dossier that documents the reproducibility of yield, impurity profile, and batch‑to‑batch consistency Worth keeping that in mind..
Concluding Remarks
The acetylation of salicylic acid with acetic anhydride provides a textbook illustration of ester formation, reaction mechanism, and practical laboratory techniques. That's why mastery of the balanced equation, stoichiometric calculations, and purification strategies equips chemists with a solid foundation for more complex synthetic challenges. Also worth noting, the safety, environmental, and scale‑up considerations highlighted here underscore the importance of responsible laboratory practice and thoughtful process design. By integrating these concepts, students and professionals alike can transition smoothly from the bench‑top synthesis of aspirin to its industrial manufacture, thereby contributing to the continued availability of one of the world’s most essential medicines Turns out it matters..