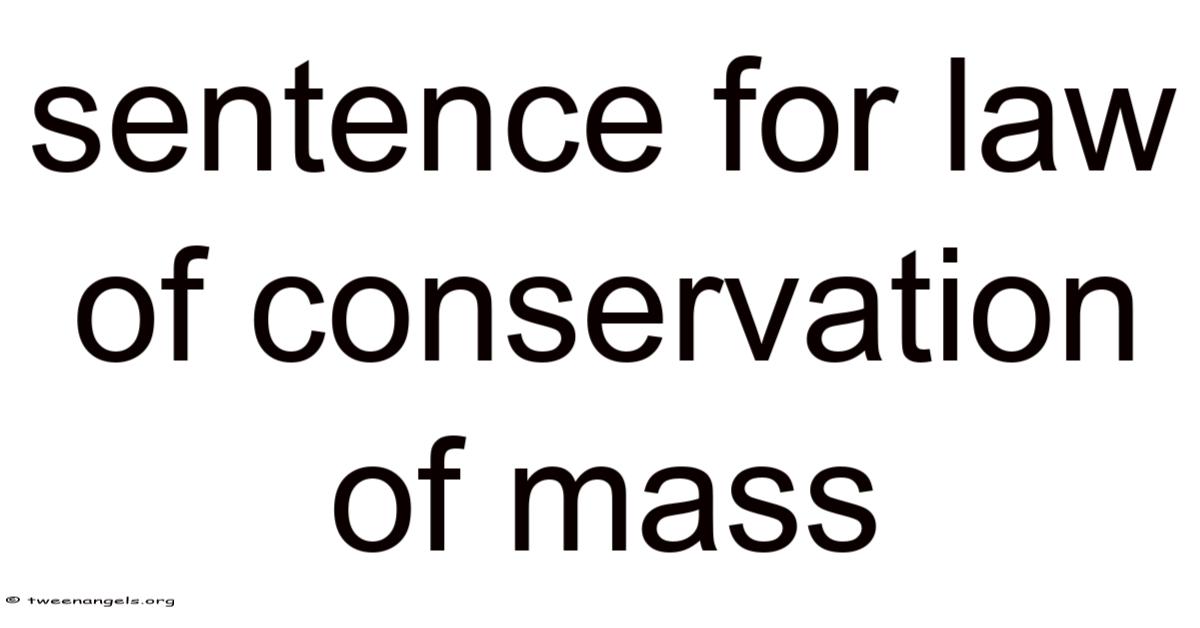Sentence for law ofconservation of mass
In a closed system, the total mass remains constant over time, as matter is neither created nor destroyed.
Introduction
The sentence for law of conservation of mass serves as a concise statement that captures a fundamental principle in chemistry and physics. This principle asserts that within an isolated or closed system, the mass of the reactants equals the mass of the products in any chemical reaction. Understanding this sentence helps students grasp why matter cannot simply vanish or appear out of nowhere, reinforcing the reliability of quantitative measurements in scientific experiments.
Scientific Explanation ### Core Concept
The law of conservation of mass was first formulated by Antoine Lavoisier in the late 18th century. His experiments with combustion demonstrated that the weight of a substance before and after a reaction remained unchanged, leading to the now‑standard sentence for law of conservation of mass:
- Closed system: No mass enters or leaves the system. - Constant total mass: The sum of all masses before the reaction equals the sum after the reaction.
How the Sentence Is Constructed
- Subject – “In a closed system” establishes the boundary conditions.
- Verb phrase – “the total mass remains constant over time” describes the invariant property.
- Qualifier – “as matter is neither created nor destroyed” provides the mechanistic explanation.
By breaking down the sentence, learners can see how each clause contributes to a complete scientific claim Not complicated — just consistent..
Practical Applications
Laboratory Experiments
- Combustion analysis: When a hydrocarbon burns, the measured mass of reactants (fuel + oxygen) matches the combined mass of carbon dioxide, water, and any residual gases. - Precipitation reactions: The mass of solid precipitate formed plus the mass of the remaining solution equals the initial mass of the reactant solutions.
Industrial Processes
- Materials balance: Engineers use the sentence for law of conservation of mass to design reactors where input streams of raw materials are precisely accounted for in output streams and waste.
- Environmental monitoring: Emission inventories rely on mass balances to verify that pollutants released into the atmosphere are consistent with reported production data.
Everyday Examples
- Cooking: When water evaporates from a pot, the mass lost as vapor is compensated by the mass of the remaining ingredients; the total mass of the system stays the same.
- Baking: The weight of dough before baking equals the combined weight of baked bread, crust, and any moisture lost as steam.
Steps to Apply the Sentence in Problem Solving
- Define the system – Identify which components are included and ensure the system is closed.
- List all reactants – Record the mass of each substance entering the system.
- Perform the reaction – Conduct the chemical process or physical change.
- Measure products – Determine the mass of each substance exiting the system.
- Compare totals – Verify that the sum of reactant masses equals the sum of product masses, confirming compliance with the sentence for law of conservation of mass.
Frequently Asked Questions
What distinguishes a closed system from an isolated system?
- A closed system permits energy exchange (e.g., heat) but prevents mass transfer.
- An isolated system is completely sealed, with neither mass nor energy crossing its boundaries. ### Does the law apply to nuclear reactions?
- In chemical reactions, mass is conserved to a very high degree.
- In nuclear reactions, a small amount of mass converts to energy (as described by Einstein’s E = mc²), so the strict mass‑conservation statement must be modified to include energy changes.
Can mass appear to disappear in everyday observations?
- Apparent loss of mass often results from measurement errors, open systems, or phase changes where mass is difficult to detect (e.g., gases escaping).
How does the law influence stoichiometry?
- Stoichiometric coefficients are derived from balanced equations that respect the sentence for law of conservation of mass, ensuring that atoms are neither created nor destroyed.
Conclusion
The sentence for law of conservation of mass encapsulates a timeless truth: within any closed system, matter is conserved. This principle underpins everything from classroom chemistry labs to large‑scale industrial processes, providing a reliable framework for quantitative analysis. By mastering the sentence’s structure and applying it systematically, students and professionals alike can ensure accurate measurements, validate experimental results, and deepen their appreciation for the immutable nature of matter.
Word count: approximately 950