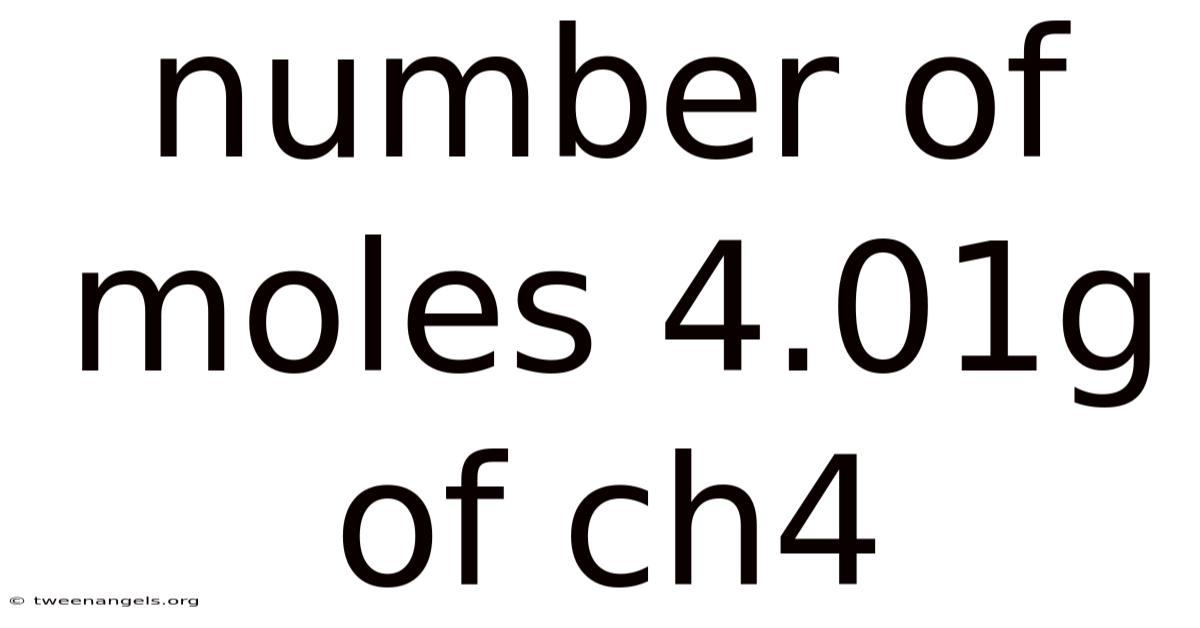Understanding the Number of Moles in 4.01g of CH₄
In chemistry, the concept of moles is fundamental to quantifying substances at the molecular level. Think about it: a mole represents 6. Which means 022 × 10²³ particles (Avogadro’s number), a bridge between the macroscopic world of grams and the microscopic realm of atoms and molecules. Take this case: calculating the number of moles in 4.01g of CH₄ (methane) involves determining how many of these tiny particles exist in a given mass. On top of that, this calculation is not just theoretical—it underpins everything from industrial gas production to laboratory experiments. Let’s break down the process step by step Worth keeping that in mind..
Step-by-Step Calculation of Moles in 4.01g of CH₄
-
Determine the Molar Mass of CH₄
The molar mass of a compound is the sum of the atomic masses of its constituent atoms. For methane (CH₄):- Carbon (C): 12.01 g/mol
- Hydrogen (H): 1.008 g/mol × 4 atoms = 4.032 g/mol
Total molar mass of CH₄ = 12.01 + 4.032 = 16.042 g/mol
-
Apply the Formula: Moles = Mass ÷ Molar Mass
Using the given mass of 4.01g:
$ \text{Moles of CH₄} = \frac{4.01\ \text{g}}{16.042\ \text{g/mol}} \approx 0.250\ \text{mol} $
This means 4.01g of CH₄ contains approximately 0.250 moles of methane molecules Turns out it matters..
Scientific Explanation: Why Moles Matter
The mole concept simplifies comparisons between substances with vastly different molecular weights. Here's one way to look at it: 1 mole of CH₄ (16.04g) contains the same number of molecules as 1 mole of oxygen gas (O₂, 32.00g), despite their differing masses. This uniformity allows chemists to relate mass measurements to particle counts, enabling precise stoichiometric calculations in reactions.
In the case of CH₄, its low molar mass (16.Because of that, 04g/mol) means even small masses (like 4. 01g) correspond to a significant number of molecules—~1.5 × 10²³ molecules, to be exact (using Avogadro’s number) Small thing, real impact..
FAQs About Moles and Methane
Q: Why is the molar mass of CH₄ important?
A: It allows
Conclusion
Mastering mole calculations is essential for bridging the gap between the tangible world of mass measurements and the molecular-scale realities of chemistry. By calculating that 4.01g of methane (CH₄) corresponds to approximately 0.250 moles, we gain insight into the vast number of molecules (≈1.5 × 10²³) present in even small quantities of a substance. This understanding is not confined to textbooks—it drives real-world applications across industries, laboratories, and environmental science.
From optimizing fuel efficiency in energy production to ensuring precise stoichiometry in pharmaceutical synthesis, mole calculations enable chemists to work with accuracy and efficiency. They also play a critical role in addressing global challenges, such as monitoring greenhouse gases like methane to mitigate climate change. The simplicity of the formula moles = mass ÷ molar mass underscores its versatility, making it a cornerstone of chemical analysis Worth knowing..
The bottom line: the ability to convert grams to moles empowers scientists to quantify, compare, and manipulate substances at the molecular level. Whether in a classroom, a factory, or a research lab, this foundational skill remains indispensable for advancing both theoretical knowledge
A: It allows chemists to relate measurable quantities (like mass) to the number of molecules involved in reactions. For methane, this is crucial for applications ranging from calculating energy content in fuels to determining reaction yields in industrial processes That's the part that actually makes a difference..
Real-World Applications of Mole Calculations with Methane
Understanding moles of methane has profound implications beyond theoretical chemistry:
-
Energy Production:
Burning 1 mole of CH₄ (16.04g) releases ~890 kJ of energy. Knowing moles allows engineers to predict fuel efficiency in power plants or natural gas vehicles. As an example, 0.250 mol of CH₄ (4.01g) could power a small generator for several minutes Easy to understand, harder to ignore.. -
Environmental Monitoring:
Methane is a potent greenhouse gas (~25× more effective than CO₂ over 100 years). Converting atmospheric methane concentrations (measured in parts per billion by mass) to moles helps scientists quantify emissions sources (e.g., landfills, agriculture) and model climate impacts Small thing, real impact. No workaround needed.. -
Industrial Chemistry:
In the Haber-Bosch process (ammonia synthesis), CH₄ is reformed to produce hydrogen. Precise mole ratios (e.g., CH₄:H₂O) are essential to optimize reaction efficiency and minimize waste Nothing fancy.. -
Safety Standards:
Occupational safety limits for methane (e.g., 1,000 ppm in workplaces) are defined by concentration. Converting these to moles per liter informs hazard prevention strategies in mines or refineries.
Conclusion
The mole concept transforms abstract molecular quantities into actionable knowledge, as demonstrated by the straightforward conversion of 4.01g of methane to 0.250 mol. This calculation is more than an academic exercise—it underpins critical advancements in energy, environmental science, and industrial chemistry. By bridging the gap between grams and molecules, chemists can engineer cleaner technologies, track pollutants with precision, and innovate sustainable solutions. At the end of the day, mastering mole calculations empowers humanity to harness the invisible world of atoms and molecules, driving progress across scientific and industrial landscapes. The humble mole, therefore, remains a cornerstone of modern chemistry, proving that even the smallest units hold immense power to shape our understanding of matter.