Insulin ______ Blood K Levels By Stimulating ______ In Cells.
tweenangels
Mar 14, 2026 · 8 min read
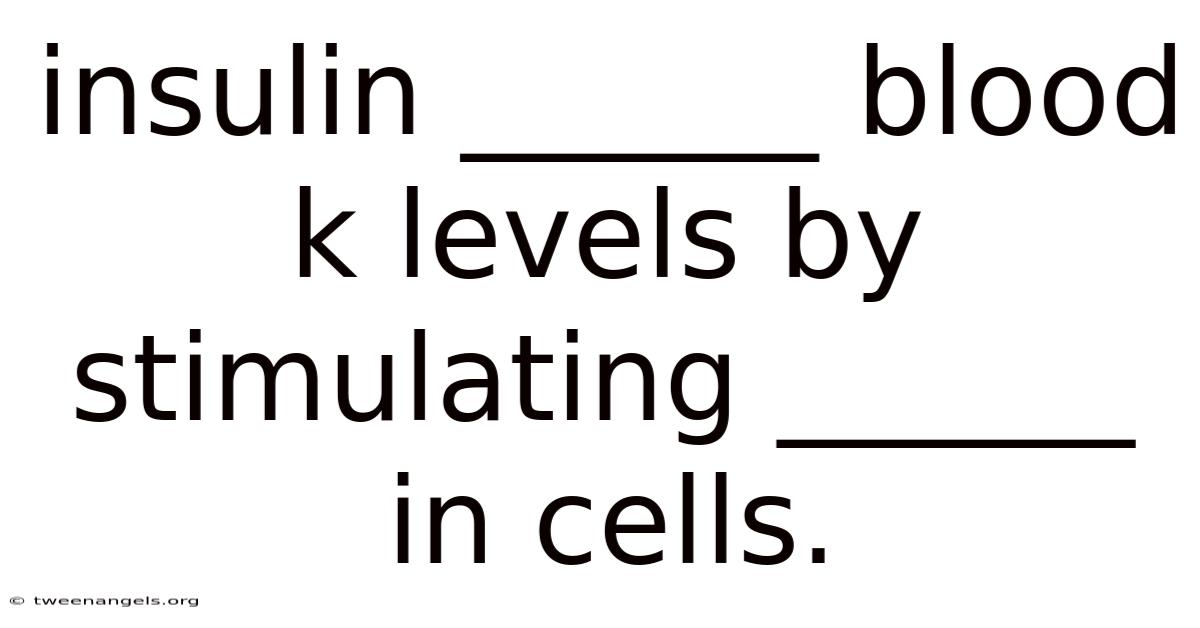
Table of Contents
Insulin Lowers Blood Potassium Levels by Stimulating Sodium-Potassium Pumps in Cells
When we think of insulin, the first thing that comes to mind is blood sugar control. This vital hormone, produced by the pancreas, is famously known for ushering glucose from the bloodstream into our cells to provide them with energy. However, insulin has another critical, life-saving function that operates in parallel: the regulation of potassium, an essential electrolyte. The precise mechanism is that insulin lowers blood potassium levels by stimulating the activity of sodium-potassium pumps (Na+/K+ ATPase) in the cell membranes of skeletal muscle and other tissues. This action drives potassium from the extracellular fluid (your blood) into the intracellular space (inside your cells), a process fundamental to maintaining the delicate electrolyte balance your heart, nerves, and muscles depend on.
The Hidden Role of Insulin in Potassium Homeostasis
Potassium is the major positively charged ion inside your body’s cells. Its concentration gradient—high inside cells, low outside—is a cornerstone of cellular physiology, powering nerve impulses, muscle contractions (including your heartbeat), and maintaining cellular fluid balance. The sodium-potassium pump is the molecular machine responsible for establishing and maintaining this gradient. It actively transports three sodium ions out of the cell and two potassium ions into the cell, using energy from ATP. This pump is constantly at work, but its activity can be modulated.
Insulin is a potent stimulator of this pump, particularly in skeletal muscle and adipose (fat) tissue, which together constitute a massive reservoir for potassium. When insulin binds to its receptor on these cells, it triggers a cascade of intracellular signals. This signaling pathway ultimately increases the number of sodium-potassium pumps inserted into the cell membrane and enhances their pumping activity. The immediate result is a rapid shift of potassium from the blood into these large cellular storage sites. This effect is separate from, but often occurs alongside, insulin’s promotion of glucose uptake. In fact, the movement of potassium into cells can be so significant that it is a standard part of the emergency treatment for hyperkalemia (dangerously high blood potassium).
Why This Mechanism Matters: Clinical Contexts
Understanding this insulin-potassium connection is not just academic; it has profound clinical implications, especially in the management of diabetes and kidney disease.
1. Diabetic Ketoacidosis (DKA) and Hyperkalemia: In untreated type 1 diabetes, a lack of insulin leads to a catastrophic state called DKA. Without insulin, glucose cannot enter cells, leading to high blood sugar. Simultaneously, the absence of insulin’s stimulatory effect on the sodium-potassium pump, combined with other factors like acidosis and lack of glucose (which normally drives potassium into cells with insulin), causes potassium to leak out of cells. This results in a total body potassium deficit (potassium is lost in urine due to high blood sugar), but paradoxically, blood potassium levels are often normal or even elevated upon hospital admission. The initiation of insulin therapy for DKA is a dual-action treatment: it lowers blood glucose and drives potassium back into cells. This can cause a rapid and dangerous drop in serum potassium levels. Therefore, potassium levels are always monitored closely during DKA treatment, and potassium is frequently added to IV fluids to prevent hypokalemia, which can cause fatal cardiac arrhythmias.
2. Treatment of Hyperkalemia: For patients with kidney failure or other conditions causing severe, life-threatening hyperkalemia, insulin (usually regular insulin) is a first-line emergency treatment. A typical dose is 5-10 units of regular insulin administered intravenously, often combined with glucose to prevent hypoglycemia. The insulin stimulates the sodium-potassium pumps, shifting potassium from the blood into muscle cells. This effect begins within 15-30 minutes, peaks at 30-60 minutes, and can lower serum potassium by 0.5-1.5 mEq/L, buying critical time while other measures (like dialysis or potassium-binding resins) take effect. This therapeutic use directly exploits the hormone’s natural physiological role.
3. The Refed Syndrome Risk: In cases of severe malnutrition or starvation, patients have adapted to a low-insulin state, with potassium largely residing outside cells. Aggressive refeeding with carbohydrates causes a surge in insulin. This sudden insulin spike, in the context of depleted total body potassium, can drive potassium rapidly into cells, leading to a precipitous and dangerous drop in blood potassium (hypokalemia). This is a classic feature of refeeding syndrome and underscores the powerful potassium-shifting effect of insulin.
The Science of the Shift: How It Happens
The stimulation of the sodium-potassium pump by insulin is a direct effect mediated by the insulin receptor’s tyrosine kinase activity. This activates downstream pathways, including the PI3K/Akt pathway, which promotes the translocation of GLUT4 glucose transporters (for glucose uptake) and also influences the trafficking and activity of the Na+/K+ ATPase pump.
It’s important to distinguish this cellular shift from renal excretion. Insulin’s primary effect on potassium is moving it between compartments (blood vs. inside cells), not directly causing the kidneys to excrete more potassium. However, by lowering blood glucose, insulin reduces the osmotic diuresis that occurs in hyperglycemia, which indirectly helps the kidneys conserve potassium and other electrolytes. The shift is also distinct from the effect of beta-2 adrenergic agonists (like albuterol), which also stimulate cellular potassium uptake but through a different receptor pathway.
Key Factors That Influence the Effect
The potency of insulin’s potassium-lowering effect is not constant and depends on several factors:
- Presence of Glucose: Insulin’s effect is significantly enhanced when glucose is available. This is why glucose is co-administered with insulin in hyperkalemia treatment—it provides the fuel for the cells and prevents hypoglycemia, but it also synergizes with insulin to promote potassium entry.
- **Beta-2 Adrenergic Activity
Beta‑2 Adrenergic Agonists: A Parallel Pathway
Although insulin is the most widely recognized hormone that drives intracellular potassium accumulation, it does not act in isolation. Catecholamines—particularly epinephrine and norepinephrine—activate β₂‑adrenergic receptors on skeletal and cardiac myocytes. This receptor stimulation also engages the G‑protein–coupled phosphoinositide‑3‑kinase (PI3K)/Akt cascade, leading to a transient up‑regulation of the Na⁺/K⁺‑ATPase and a consequent intracellular shift of potassium. In clinical practice, high‑dose inhaled or intravenous β₂‑agonists (e.g., albuterol) are frequently employed as adjunctive therapy for hyperkalemia, especially when rapid potassium reduction is needed but intravenous access is limited or contraindicated.
The magnitude of the potassium‑lowering effect produced by β₂‑agonists is generally modest compared with insulin, typically lowering serum potassium by 0.2–0.5 mEq/L over the first hour. Nevertheless, the rapid onset (within minutes) and the ability to administer these agents without concurrent glucose make them valuable in specific scenarios—such as acute asthma exacerbations where hyperkalemia co‑exists with severe bronchospasm. Clinicians must, however, be vigilant for tachyphylaxis, arrhythmias, and the potential for exacerbating underlying cardiac disease.
Renal Excretion: The Secondary, Slower Route
Beyond intracellular redistribution, insulin exerts a secondary influence on renal potassium handling. By normalizing plasma glucose, insulin diminishes the osmotic diuresis that accompanies hyperglycemia, thereby preserving the tubular reabsorption of sodium and water. This preservation indirectly supports the kidney’s ability to retain potassium when needed and to excrete it when appropriate. Moreover, insulin stimulates the activity of the Na⁺/K⁺‑ATPase in renal tubular cells, modestly enhancing distal tubular potassium secretion. This effect is most evident in states of chronic hyperglycemia, where persistent hyperinsulinemia contributes to a subtle, long‑term shift in renal potassium handling.
Clinical Pearls and Practical Considerations
- Timing Is Everything – The peak intracellular shift of potassium occurs roughly 30–60 minutes after insulin administration, underscoring the importance of monitoring serum potassium at this interval rather than relying on immediate post‑infusion values.
- Glucose Co‑Administration – Providing dextrose (usually 5–10 g per 1 U insulin) prevents hypoglycemia and ensures that target cells have sufficient substrate to engage the Na⁺/K⁺‑ATPase efficiently.
- Avoiding Over‑Correction – Aggressive insulin dosing can precipitate severe hypokalemia, especially in patients with pre‑existing potassium depletion or renal impairment. Target reductions of 0.5–1.0 mEq/L per hour are generally considered safe.
- Monitoring for Complications – Hypoglycemia, hypokalemia, and rarely, electrolyte disturbances such as hypomagnesemia, must be anticipated and managed promptly.
- Alternative Strategies – When insulin is contraindicated (e.g., in type 1 diabetic ketoacidosis with severe hyperglycemia) or ineffective, renal replacement therapy, potassium‑binding resins (patiromer, sodium zirconium cyclosilicate), or aggressive diuresis become essential components of the therapeutic arsenal.
Integrating Hormonal Knowledge into Patient Care
Understanding insulin’s potassium‑shifting mechanism equips clinicians with a mechanistic lens through which to interpret laboratory trends and therapeutic responses. Recognizing that insulin’s effect is transient, glucose‑dependent, and mediated through a well‑defined intracellular signaling cascade encourages more precise dosing, better coordination with ancillary treatments, and heightened vigilance for metabolic complications. Moreover, appreciating the interplay between insulin, β₂‑adrenergic signaling, and renal potassium handling fosters a holistic approach to electrolyte management—one that aligns hormonal physiology with clinical pragmatism.
Conclusion
Insulin is a pivotal regulator of serum potassium, wielding its influence primarily through stimulation of the Na⁺/K⁺‑ATPase and secondarily through modulation of renal handling. This dual action enables clinicians to harness insulin as a rapid, glucose‑dependent tool for lowering serum potassium in hyperkalemic emergencies, while also appreciating its role within broader metabolic contexts such as refeeding syndrome. By integrating insights from hormone physiology, bedside monitoring, and adjunctive therapies, healthcare providers can navigate the delicate balance between correcting hyperkalemia and avoiding the pitfalls of over‑correction, ultimately delivering safer, more effective care for patients whose potassium homeostasis is compromised.
Latest Posts
Latest Posts
-
Which Of The Following Occurs During Expiration
Mar 14, 2026
-
How To Find Z Score Using Ti 84
Mar 14, 2026
-
Determine The Following Indefinite Integral Check Your Work By Differentiation
Mar 14, 2026
-
How Would You Make The Following Compounds From N Benzylbenzamide
Mar 14, 2026
-
A Walking Stick Insect Blends In With Its Environment
Mar 14, 2026
Related Post
Thank you for visiting our website which covers about Insulin ______ Blood K Levels By Stimulating ______ In Cells. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.