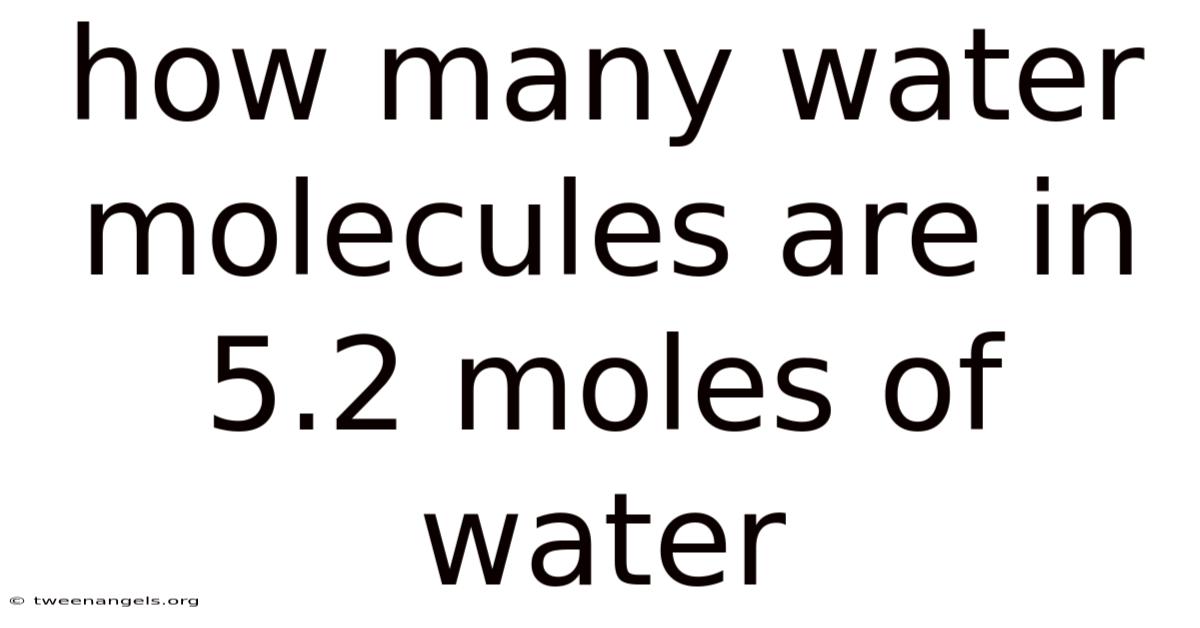How Many Water Molecules Are in 5.2 Moles of Water?
Understanding how many water molecules are in 5.2 moles of water requires a dive into the fascinating world of stoichiometry and the fundamental constants of chemistry. For many students, the concept of a "mole" can feel abstract or overwhelming, but it is essentially just a chemist's version of a "dozen." Just as a dozen always means 12 items, a mole always represents a specific, massive number of particles. By applying Avogadro's number, we can bridge the gap between the macroscopic world we see in a beaker and the microscopic world of individual molecules Worth keeping that in mind..
Introduction to the Mole Concept
In chemistry, dealing with individual atoms or molecules is practically impossible because they are far too small to count one by one. That said, to solve this, scientists use the mole (mol) as a standard unit of measurement. The mole is the SI unit for the amount of substance Not complicated — just consistent. Which is the point..
The magic number that connects moles to actual particles is known as Avogadro's number. Named after Amedeo Avogadro, this constant is approximately:
$6.022 \times 10^{23}$
Basically, 1 mole of any substance—whether it is gold atoms, oxygen molecules, or water molecules—contains exactly $6.022 \times 10^{23}$ representative particles. Worth adding: when we ask how many molecules are in 5. 2 moles of water, we are essentially asking for the total count of particles contained within 5.2 "chemist's dozens.
Step-by-Step Calculation: Finding the Number of Molecules
To calculate the number of molecules in a given amount of moles, you can follow a simple mathematical formula. Here is the breakdown of the process for 5.2 moles of water ($\text{H}_2\text{O}$).
1. Identify the Known Values
First, list the information provided in the problem:
- Amount of substance (n): $5.2\text{ moles}$
- Avogadro's Number ($N_A$): $6.022 \times 10^{23}\text{ molecules/mol}$
2. Apply the Formula
The formula to find the total number of particles ($N$) is: $\text{Number of Particles} = \text{Moles} \times \text{Avogadro's Number}$ $N = n \times N_A$
3. Perform the Multiplication
Now, plug the values into the equation: $N = 5.2\text{ mol} \times (6.022 \times 10^{23}\text{ molecules/mol})$
When multiplying, you multiply the coefficients (5.Because of that, 2 and 6. 022) and keep the power of ten: $5.2 \times 6.022 = 31.
So, the result is: $31.3144 \times 10^{23}\text{ molecules}$
4. Convert to Standard Scientific Notation
In scientific notation, we typically keep only one non-zero digit before the decimal point. To do this, we move the decimal one place to the left and increase the exponent by one: $3.13144 \times 10^{24}\text{ molecules}$
5. Rounding for Significant Figures
Since the initial value "5.2" has two significant figures, our final answer should reflect that precision: $3.1 \times 10^{24}\text{ molecules}$
Final Answer: There are approximately $3.1 \times 10^{24}$ water molecules in 5.2 moles of water.
Scientific Explanation: Why This Number is So Large
It is often difficult for the human mind to visualize a number as large as $3.1 \times 10^{24}$. Think about it: to put this in perspective, if every water molecule in 5. 2 moles of water were a single grain of sand, you would have enough sand to cover the entire Earth in a layer several meters deep No workaround needed..
This massive scale is necessary because molecules are incredibly tiny. So a single molecule of water consists of two hydrogen atoms and one oxygen atom bonded together. Because they are so small, it takes a staggering amount of them to create a substance we can actually see, touch, and measure in a laboratory.
Some disagree here. Fair enough Worth keeping that in mind..
The Relationship Between Moles, Mass, and Molecules
While we focused on the number of molecules, it is helpful to understand how this relates to the actual weight of the water. To find the mass of 5.2 moles of water, we use the molar mass.
- Molar mass of $\text{H}_2\text{O}$: $\approx 18.015\text{ g/mol}$
- Mass calculation: $5.2\text{ mol} \times 18.015\text{ g/mol} \approx 93.68\text{ grams}$
This reveals a striking truth: a relatively small amount of water—less than 100 grams (about 93 ml)—contains over three septillion molecules.
Common Pitfalls to Avoid
When students tackle these problems, they often run into a few common errors. Being aware of these can help you avoid mistakes in your chemistry assignments:
- Confusing Moles with Molecules: Remember that a "mole" is the group, and a "molecule" is the individual unit. Never leave your answer as "5.2" when the question asks for the number of molecules.
- Calculator Errors with Exponents: When typing $6.022 \times 10^{23}$ into a calculator, always use the EXP or EE button. If you type it as
6.022 * 10 ^ 23, ensure you use parentheses correctly, or the calculator might perform the operations in the wrong order. - Ignoring Significant Figures: In science, the precision of your answer depends on the precision of your measurements. If the problem gives you "5.2" (two sig figs), providing an answer with ten decimal places is scientifically inaccurate.
FAQ: Frequently Asked Questions
What is the difference between a mole of atoms and a mole of molecules?
A mole of atoms contains $6.022 \times 10^{23}$ individual atoms. A mole of molecules contains $6.022 \times 10^{23}$ molecules. On the flip side, since one water molecule ($\text{H}_2\text{O}$) contains three atoms (2 Hydrogen, 1 Oxygen), one mole of water molecules actually contains three moles of atoms Worth knowing..
Does the number of molecules change if the water is liquid, ice, or steam?
No. Whether the water is a solid, liquid, or gas, the number of molecules remains the same as long as the amount of substance (5.2 moles) does not change. Only the distance between the molecules and their energy levels change during a phase shift.
Why is Avogadro's number so specific?
Avogadro's number is defined based on the mass of carbon-12. It is the number of atoms in exactly 12 grams of carbon-12, which provides a universal bridge between the atomic mass unit (amu) and the gram.
Conclusion
Calculating the number of molecules in 5.Practically speaking, 2 moles of water is a straightforward process once you understand the role of Avogadro's number. By multiplying the number of moles by $6.022 \times 10^{23}$, we discover that 5.On the flip side, 2 moles of water contains a staggering $3. 1 \times 10^{24}$ molecules.
This exercise highlights the incredible scale of the molecular world. Still, it teaches us that the substances we interact with every day are composed of an almost unimaginable number of tiny particles, all working together according to the laws of chemistry. Mastering this conversion is a foundational step for anyone studying chemistry, as it allows for the precise calculation of reactants and products in chemical reactions Small thing, real impact. Practical, not theoretical..
Understanding the intricacies of your chemistry assignments requires a careful approach to each calculation, especially when dealing with large quantities and precise conversions. Think about it: for instance, when working with Avogadro’s number, it’s crucial to remember that 1 mole equals $6. 022 \times 10^{23}$ entities, whether those are atoms, molecules, or even ions. A common challenge lies in distinguishing between moles and the actual number of molecules, ensuring that you apply the correct scientific conventions. This distinction prevents simple errors, such as miscalculating the transformation from moles to molecules Easy to understand, harder to ignore..
And yeah — that's actually more nuanced than it sounds.
When performing computations, particularly with exponents or significant figures, precision becomes very important. A misstep in typing syntax or overlooked sig figs can distort your results significantly. As an example, if you mistakenly use decimal notation instead of scientific, the calculator’s interpretation might change entirely, leading to incorrect answers. Always double-check your steps and align your units consistently.
Another key point to keep in mind is how phase changes affect molecular count. In real terms, while the total number of molecules remains constant, their physical state alters the spacing and arrangement—this is why solid, liquid, and gas phases each have their own molecular behaviors. This subtlety reinforces the importance of context in your work.
The short version: each challenge in your chemistry tasks reinforces the value of accuracy and understanding. By mastering these concepts, you build a stronger foundation for tackling complex problems Nothing fancy..
Conclusion: Navigating the nuances of chemistry assignments sharpens your analytical skills and deepens your appreciation for the microscopic universe behind everyday substances. Stay precise, stay curious, and let these lessons guide your future studies.