How Many Significant Numbers Are In 10.0
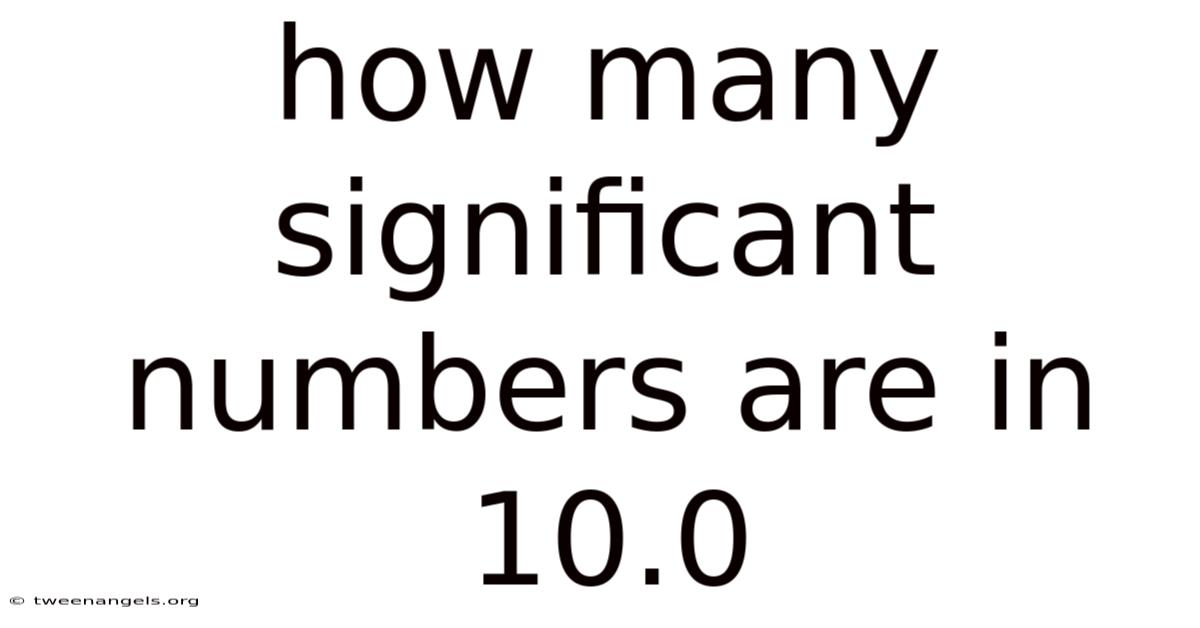
How Many Significant Figures Are in 10.0? A Deep Dive into Precision
The number 10.0 appears simple, almost trivial. Yet, within its three digits lies a fundamental concept of science, engineering, and data integrity: significant figures. The answer to "how many significant figures are in 10.0?" is definitively three. This seemingly small distinction between "10" and "10.0" represents a world of difference in measured precision. Understanding why requires exploring the rules that govern significant figures and the profound message that a single trailing zero after a decimal point conveys about the certainty of a measurement.
The Core Rules: Why 10.0 Has Three Significant Figures
Significant figures are the digits in a number that carry meaning contributing to its precision. They include all certain digits plus the first uncertain (or estimated) digit. The rules for identifying them are systematic:
- All non-zero digits are significant. (e.g., 123 has 3 sig figs).
- Zeros between non-zero digits are significant. (e.g., 101 has 3 sig figs).
- Leading zeros (zeros before the first non-zero digit) are not significant. They only locate the decimal point. (e.g., 0.0025 has 2 sig figs).
- Trailing zeros (zeros after the last non-zero digit) are significant only if the number contains a decimal point. This is the critical rule for 10.0.
Applying these rules to 10.0:
- The digit '1' is non-zero and significant.
- The digit '0' is between the '1' and the decimal point? No, it's after the '1' and before the decimal. Wait, let's parse it correctly: The number is 1, then a 0, then a decimal point, then another 0. The structure is
10.0. - The first '0' is a trailing zero before the decimal point? Actually, in 10.0, the digits are: '1' (non-zero), '0' (a trailing zero to the left of the decimal), and '0' (a trailing zero to the right of the decimal).
- Rule 4 states: trailing zeros are significant if the number contains a decimal point. Since 10.0 explicitly has a decimal point, both zeros are significant.
- Therefore: 1 (significant) + 0 (significant) + 0 (significant) = 3 significant figures.
This contrasts sharply with the number 10, which has two significant figures. The absence of a decimal point means the trailing zero is ambiguous—it could be a placeholder or an estimated digit. Without the decimal, we assume it is not significant. Writing 10.0 removes that ambiguity, declaring: "This measurement is precise to the tenths place."
The Scientific Explanation: What the Decimal Point Communicates
The decimal point is not just a punctuation mark; it is a declaration of precision. In experimental science and engineering, every measurement has an inherent uncertainty. When you record a value, you are stating what you are certain about and what you are estimating.
- 10 implies the measurement is certain to the ones place. The true value could reasonably be anywhere between 9.5 and 10.5. The '0' is merely a placeholder; the measurer did not estimate the tenths place.
- 10.0 implies the measurement is certain to the tenths place. The true value is believed to be between 9.95 and 10.05. The measurer looked at the instrument (e.g., a ruler with millimeter markings, a digital caliper) and could confidently estimate one digit beyond the smallest marked division. That final '0' is the estimated digit, and it is meaningful. It says, "I am sure the value is 10, and I am even confident that it is not 10.1 or 9.9."
This distinction has real consequences. In calculations, the number of significant figures in your data dictates the number you can reliably report in your result. Using 10 (2 sig figs) versus 10.0 (3 sig figs) in a formula will yield answers with different implied precision. Propagating error through calculations depends entirely on these initial precision declarations.
Illustrative Examples: From Ambiguity to Clarity
Let's solidify this with a series of comparisons:
- 100 – Has 1 significant figure (the '1'). The two zeros are trailing placeholders without a decimal. The measurement is precise only to the hundreds place (~50 to 150).
- 100. – Has 3 significant figures. The decimal point makes both trailing zeros significant. Precision is to the ones place (99.5 to 100.5).
- 100.0 – Has 4 significant figures. Precision is to the tenths place (99.95 to 100.05).
- 0.0100 – Has 3 significant figures. The leading zeros are not significant. The trailing zeros after the decimal are significant. This measures a very small quantity with precision to the hundred-thousandths place.
- 10.0 – Has 3 significant figures. The decimal point validates both the zero in the ones place and the zero in the tenths place as meaningful.
The progression 10 → 10.0 → 10.00 tells a clear story of increasing confidence and instrument resolution. Each additional digit after the decimal represents a tenfold increase in the claimed precision of the original measurement.
Common Mistakes and Pitfalls
Misunderstanding 10.0 often stems from two errors:
- Assuming the trailing zero is insignificant. Some treat "10.0" as merely a stylistic variant of "10," failing to recognize that the decimal point and the final zero actively declare a higher precision. This erases the measurer's extra effort and judgment.
- Overestimating precision from calculation. Students often report a result like "10.0" after multiplying two numbers each with only two significant figures (e.g., 3.1 × 3.2 = 9.92). The product cannot logically have three significant figures; the true precision is two (10). The estimated digit must be dropped to avoid false precision.
These errors corrupt data integrity. Reporting "10.0" for a measurement only precise to ±0.5 is not modesty—it is scientific dishonesty. Conversely, reporting "10" for a measurement made with a micrometer that reads to 0.01 mm wastes valuable information and understates the experiment's capability.
The Real-World Stakes
This is not pedantry. In pharmaceutical dosing, a concentration written as "5.0 mg/mL" versus "5 mg/mL" could represent a 2% difference—critical for a narrow therapeutic index drug. In aerospace engineering, specifying a turbine blade length as "10.0 cm" versus "10 cm" might determine whether it fits within a tolerance of ±0.05 cm or a looser ±0.5 cm. The former requires a more expensive, precise manufacturing process. In climate science, a trend reported as "+0.10°C per decade" (two sig figs) carries different weight than "+0.100°C per decade" (three sig figs); the latter suggests a much higher certainty in the instrumental record.
Significant figures are the grammar of quantitative communication. They force the writer to confront uncertainty and the reader to interpret numbers with appropriate skepticism. They transform raw digits into a statement about the reliability of knowledge.
Conclusion
Ultimately, the distinction between "10" and "10.0" is a profound one. It is the difference between saying, "I know this is about ten," and saying, "I have measured this, and I am confident it is not 9.9 or 10.1." The former is an estimate; the latter is a measurement. Mastering this convention is not about adhering to arbitrary rules, but about cultivating a disciplined mindset that respects the limits of observation. In science and engineering, how you write a number is inseparable from what you know—and what you do not know. Precision, after all, is not just a property of the instrument; it is a commitment to intellectual honesty, encoded in a single, silent decimal point.
Latest Posts
Latest Posts
-
Introduction To Java Programming And Data Structures 12th Edition
Mar 22, 2026
-
Planning And Administering Early Childhood Programs
Mar 22, 2026
-
General Organic And Biological Chemistry 4th Edition
Mar 22, 2026
-
Midpoint Formula Of Price Elasticity Of Demand
Mar 22, 2026
-
Label The Following Fatty Acids As Saturated Or Unsaturated
Mar 22, 2026