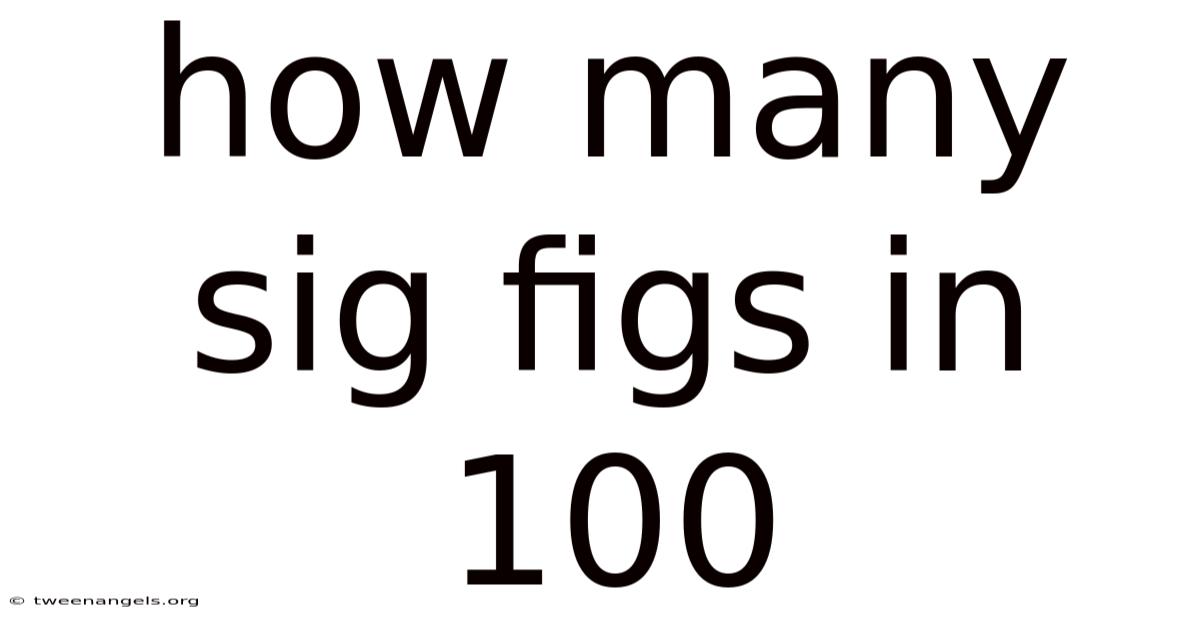How Many Sig Figs in 100? A Detailed Exploration of Significant Figures in Numbers
When dealing with numerical data, especially in scientific or mathematical contexts, understanding the concept of significant figures is crucial. Significant figures, often abbreviated as "sig figs," refer to the digits in a number that convey meaningful information about its precision. Which means they help communicate the reliability of a measurement or calculation. Now, a common question that arises in this context is: *How many sig figs are in 100? In practice, * This seemingly simple question can be nuanced, depending on how the number is presented and the context in which it is used. In this article, we will break down the rules governing significant figures, apply them to the number 100, and explore why this topic matters in real-world applications.
What Are Significant Figures?
Before answering the specific question about 100, Make sure you define what significant figures are. 3 cm, it has three significant figures. Even so, it matters. Significant figures are the digits in a number that are known with certainty plus one final digit that is estimated. They indicate the precision of a measurement or calculation. Here's one way to look at it: if a ruler measures a length as 12.This precision is critical in fields like chemistry, physics, and engineering, where even small differences in measurements can have significant impacts.
The rules for determining significant figures are not always straightforward, but they are consistent. These rules help check that numbers are interpreted correctly, especially when performing calculations or comparing data. The key is to distinguish between digits that are meaningful and those that are placeholders.
Rules for Determining Significant Figures
To answer the question how many sig figs in 100, we must first understand the general rules for identifying significant figures. These rules apply to all numbers, whether they are whole numbers, decimals, or scientific notation. Here are the key guidelines:
-
Non-zero digits are always significant.
Any digit from 1 to 9 is considered significant. As an example, in the number 456, all three digits are significant. -
Zeros between non-zero digits are significant.
Zeros that appear between two non-zero digits are always significant. Take this case: in 101, all three digits are significant Less friction, more output.. -
Leading zeros are not significant.
Zeros that appear before the first non-zero digit are not significant. They are used only to indicate the position of the decimal point. As an example, in 0.0045, only the 4 and 5 are significant. -
Trailing zeros in a whole number with no decimal point are not significant.
This is a critical rule when analyzing the number 100. Trailing zeros (zeros at the end of a number) are not considered significant unless there is a decimal point. As an example, 100 has one significant figure, while 100. has three. -
Trailing zeros in a decimal number are significant.
If a number has a decimal point, trailing zeros after the decimal are significant. Take this: 100.0 has four significant figures. -
Scientific notation clarifies significance.
Numbers in scientific notation explicitly show the number of significant figures. Take this: 1.00 × 10² has three significant figures, while 1 × 10² has only one.
These
These rules for determining significant figures are fundamental to accurate scientific communication and data analysis. Day to day, understanding and applying them consistently prevents misinterpretations and ensures the reliability of experimental results and calculations. While the rules can seem complex at first, practicing with various examples solidifies comprehension.
So, to directly address the question of how many significant figures are in 100, we apply rule number four: trailing zeros in a whole number with no decimal point are not significant. Because of this, the number 100 has two significant figures. The 1 represents the first non-zero digit, and the 0 represents the second. On top of that, this is a crucial detail, particularly when comparing results or performing calculations involving numbers with varying degrees of precision. Ignoring the significance of trailing zeros can lead to erroneous conclusions.
To wrap this up, determining significant figures is a vital skill in any scientific discipline. And the seemingly simple number 100 provides a clear illustration of this principle, highlighting the importance of paying close attention to the digits and their context when interpreting and communicating scientific information. By understanding and correctly applying the rules, we can ensure the accuracy and reliability of our work. Mastering this concept is a cornerstone of rigorous scientific practice.
These rules for determining significant figures are fundamental to accurate scientific communication and data analysis. Understanding and applying them consistently prevents misinterpretations and ensures the reliability of experimental results and calculations. While the rules can seem complex at first, practicing with various examples solidifies comprehension.
So, to directly address the question of how many significant figures are in 100, we apply rule number four: trailing zeros in a whole number with no decimal point are not significant. Which means, the number 100 has two significant figures. In practice, the 1 represents the first non-zero digit, and the 0 represents the second. This is a crucial detail, particularly when comparing results or performing calculations involving numbers with varying degrees of precision. Ignoring the significance of trailing zeros can lead to erroneous conclusions.
To wrap this up, determining significant figures is a vital skill in any scientific discipline. Mastering this concept is a cornerstone of rigorous scientific practice. By understanding and correctly applying the rules, we can ensure the accuracy and reliability of our work. Consistent application of these rules ensures that scientific findings are communicated with precision and avoid misleading implications. On the flip side, the seemingly simple number 100 provides a clear illustration of this principle, highlighting the importance of paying close attention to the digits and their context when interpreting and communicating scientific information. **Beyond simple calculations, the concept of significant figures extends to data analysis, statistical interpretations, and the presentation of results. The bottom line: accurate reporting of significant figures is not just about adhering to a set of rules; it's about upholding the integrity of scientific inquiry and maintaining a clear and honest representation of the data That's the part that actually makes a difference. Nothing fancy..
Applying Significant Figures in Real‑World Contexts
When we move beyond textbook problems and into the laboratory or fieldwork, the abstract rules for significant figures become concrete tools that shape every decision we make—from instrument selection to data reporting.
1. Instrument Precision and Calibration
Every measuring device has a specified resolution—the smallest increment it can reliably display. 30 V**, the trailing zero after the decimal point is significant because the instrument’s design guarantees that level of precision. Conversely, if a ruler is marked in millimetres, a length measured as 45 mm contains only two significant figures; writing **45.For a digital voltmeter that reads to the nearest 0.Here's the thing — 01 V, the last digit is inherently uncertain, and any value reported should reflect that limitation. Plus, if the voltmeter displays 12. 0 mm would imply a precision the tool cannot provide Easy to understand, harder to ignore..
2. Propagation of Uncertainty
In multi‑step calculations, the rule of thumb is to retain the fewest significant figures present in any intermediate result. As an example, consider the computation of the density of a metal sample:
[ \rho = \frac{m}{V} ]
- Mass, (m = 12.5\ \text{g}) (three significant figures)
- Volume, (V = 2.00\ \text{cm}^3) (three significant figures)
Carrying out the division yields (\rho = 6.25\ \text{g/cm}^3). Which means since both inputs have three significant figures, the final answer should be rounded to three, giving 6. So 25 g·cm⁻³. If, however, the volume were measured as 2 cm³ (one significant figure), the density must be reported as 6 g·cm⁻³ to avoid implying a false level of certainty Most people skip this — try not to. Nothing fancy..
3. Reporting Results in Publications
Scientific journals often require authors to include a statement of uncertainty alongside each measured value, typically in the format “(x \pm \sigma)”. 03**. If an experiment yields (x = 3.03), the uncertainty has two significant figures; therefore, the result should be reported as **3.1416) with an uncertainty of (\pm 0.But 14 ± 0. In real terms, the number of significant figures in the uncertainty dictates how many digits should be retained in the main value. This practice prevents the illusion of precision that could mislead readers.
4. Statistical Analyses
When performing statistical tests, the precision of input data directly influences the confidence intervals and p‑values. Conversely, under‑reporting precision can mask genuine trends. That said, over‑stating significant figures in raw data can artificially narrow confidence intervals, leading to over‑confident conclusions. A balanced approach—honoring the true measurement limits—maintains the integrity of statistical inference.
Common Pitfalls and How to Avoid Them
| Pitfall | Why It Happens | Remedy |
|---|---|---|
| Assuming all zeros are significant | Misinterpretation of trailing zeros in whole numbers | Use scientific notation (e.Plus, g. , (1. |
A Quick Checklist for Researchers
- Identify the precision of each measurement tool.
- Write numbers in scientific notation when ambiguity exists.
- Carry extra digits through calculations; round only at the end.
- Match the number of significant figures in the final answer to the least‑precise input.
- State uncertainties explicitly and let them dictate the reported precision.
By habitually following this checklist, researchers embed rigor into every stage of data handling.
Final Thoughts
The journey from a simple integer like 100 to the complex datasets of modern science underscores a fundamental truth: significant figures are not merely a pedagogical exercise; they are the language of precision. Whether you are measuring the concentration of a solution, the speed of a particle, or the luminosity of a distant star, the way you express numbers conveys how much you truly know—and, equally importantly, how much you do not know.
In practice, this means:
- Respecting the limits of your instruments,
- Communicating uncertainty transparently, and
- Applying the rules consistently across all calculations.
When these principles are internalized, the scientific community benefits from data that are comparable, reproducible, and trustworthy. The seemingly modest decision of whether the trailing zeros in “100” are significant becomes a microcosm of a broader commitment to methodological integrity.
In conclusion, mastering significant figures equips scientists with a vital tool for accurate measurement, reliable computation, and honest reporting. By integrating these concepts into everyday laboratory work and scholarly communication, we uphold the standards that allow science to progress on a foundation of clarity and precision That's the part that actually makes a difference. And it works..