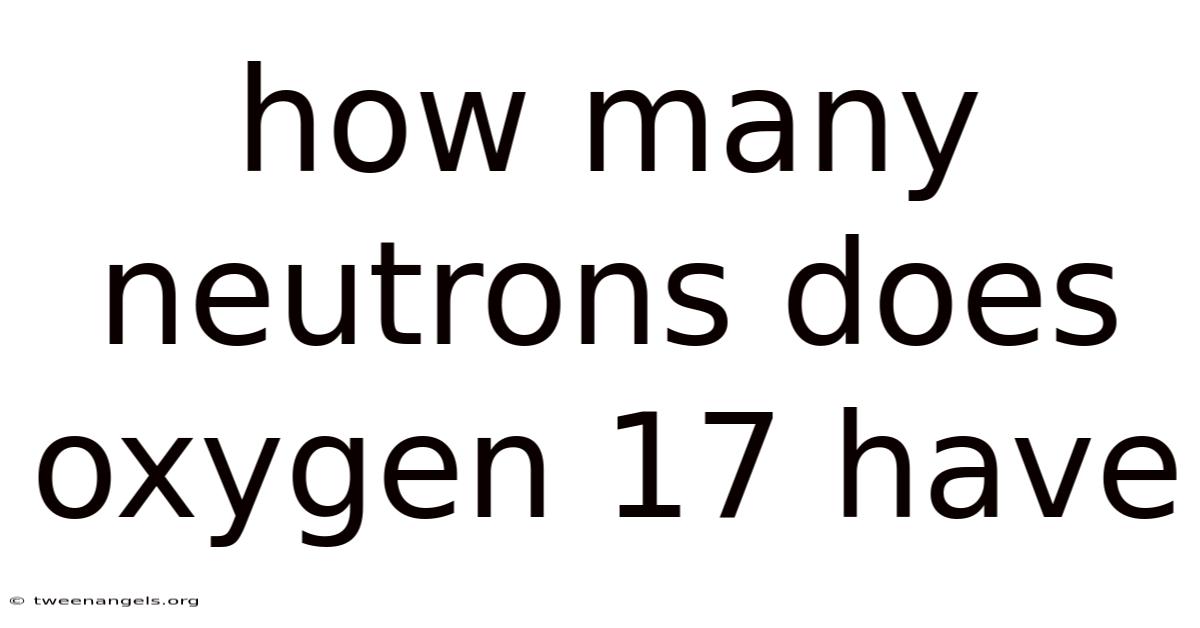Oxygen-17, a stable isotope of the element oxygen, possesses 9 neutrons. And this specific number is derived from a fundamental relationship within the atom's nucleus and defines the isotope's unique properties and scientific utility. To understand why oxygen-17 has exactly 9 neutrons, one must first grasp the concepts of atomic number, mass number, and isotopes.
Understanding Atomic Structure: The Key to Neutron Count
Every atom is built around a nucleus containing protons and neutrons, surrounded by a cloud of electrons. That said, two critical numbers define an atom's identity and variant forms:
- Atomic Number (Z): This is the number of protons in the nucleus. For all oxygen atoms, regardless of the isotope, the atomic number is 8. But this is what makes the atom chemically oxygen; it determines the element's position on the periodic table and its chemical behavior. * Mass Number (A): This is the total number of protons and neutrons combined in the nucleus. It is a whole number that approximates the atom's mass. The number after the element name—in this case, "17" in oxygen-17—is the mass number.
The calculation for the number of neutrons (N) is straightforward: Neutrons (N) = Mass Number (A) - Atomic Number (Z)
For oxygen-17: N = 17 - 8 = 9 neutrons Easy to understand, harder to ignore..
This simple equation is the universal key to determining neutron count for any isotope.
The Family of Oxygen: A Tale of Three Stable Isotopes
Oxygen is unique among common elements in having three stable, naturally occurring isotopes. * Oxygen-18 (¹⁸O): The second most abundant stable isotope (~0.76%). Its odd neutron number gives it a non-zero nuclear spin, a property crucial for its primary scientific applications. Which means * Oxygen-16 (¹⁶O): The most abundant isotope (>99. It is a "doubly magic" nucleus, meaning it has both a magic number of protons (8) and neutrons (8), contributing to its exceptional stability and abundance. 20%). On top of that, it has 8 protons and 9 neutrons (17 - 8 = 9). * Oxygen-17 (¹⁷O): A rare, stable isotope (~0.That said, 04% natural abundance). And it has 8 protons and 8 neutrons (16 - 8 = 8). Their differences lie solely in their neutron count, which affects their atomic mass and certain nuclear properties. It has 8 protons and 10 neutrons (18 - 8 = 10) That's the part that actually makes a difference..
The existence of these isotopes demonstrates that atoms of the same element can have different masses. This variation is not a chemical difference but a nuclear one, stemming from the number of neutrons in the nucleus Not complicated — just consistent..
Why Does Oxygen-17 Have 9 Neutrons? The Nuclear Perspective
Neutrons play a vital role in the nucleus. They provide the strong nuclear force necessary to bind the positively charged protons together, overcoming the protons' electrostatic repulsion. The specific number of neutrons in a stable isotope is not arbitrary; it represents a configuration where the nuclear forces are balanced, creating a stable or long-lived nucleus.
For lighter elements like oxygen, the most stable isotopes typically have roughly equal numbers of protons and neutrons (N ≈ Z). So oxygen-16 (8p, 8n) perfectly fits this pattern. Oxygen-17 (8p, 9n) has one extra neutron. That's why this slight neutron excess still results in a stable configuration, but its odd neutron number breaks the symmetry, giving the nucleus a net spin of 5/2. This spin is the source of its most important property: it is NMR-active Small thing, real impact..
The Scientific Significance of Oxygen-17's Neutron Count
The fact that oxygen-17 has 9 neutrons—an odd number—is not just a trivia fact. It is the reason this isotope is a powerhouse in scientific research, particularly in the field of Nuclear Magnetic Resonance (NMR) spectroscopy Simple, but easy to overlook..
-
NMR Spectroscopy: Nuclei with an odd number of protons and/or neutrons possess a property called nuclear spin. Oxygen-17 has a spin of 5/2. When placed in a strong magnetic field, these spinning nuclei can absorb and re-emit radiofrequency energy. This allows scientists to probe the local chemical environment of oxygen atoms in molecules. While its low natural abundance (0.04%) makes detection challenging, enriching samples with oxygen-17 provides unparalleled insight into:
- The structure of complex molecules like proteins, nucleic acids, and polymers.
- Reaction mechanisms in chemistry and biochemistry by tracking oxygen atoms.
- The dynamics of water molecules in biological systems and materials.
-
Geochemistry and Paleoclimatology: The ratio of oxygen-18 to oxygen-16 (δ¹⁸O) in ice cores, marine sediments, and fossilized shells is a fundamental proxy for past temperatures and ice volume. Oxygen-17 (δ¹⁷O) measurements provide a secondary, complementary check on these records and can help correct for certain non-temperature-related effects. The distinct mass of oxygen-17 (due to its 9 neutrons) means it behaves slightly differently during physical processes like evaporation and condensation compared to oxygen-16 and oxygen-18.
-
Medical Research: While not used as a direct tracer like carbon-13 or nitrogen-15 due to cost and detection challenges, oxygen-17-enriched water (H₂¹⁷O) is used in specialized research to measure blood flow and metabolic rates in the brain and other organs via NMR techniques.
How to Determine Neutrons for Any Isotope: A Step-by-Step Guide
The process used for oxygen-17 applies universally. Note its atomic number (Z), which is the number of protons. It represents protons + neutrons. But Number of Neutrons = Mass Number (A) - Atomic Number (Z) 4. 2. Consider this: for oxygen, Z = 8. Identify the Mass Number (A): This is the superscript number preceding or following the element symbol (e.Because of that, follow these steps for any isotope:
- So naturally, Identify the Element: Find the element on the periodic table. Apply the Formula: Subtract the atomic number from the mass number. 3. g.In real terms, , the "17" in ¹⁷O). Example with Oxygen-18: A = 18, Z = 8.
Neutrons = 18 – 8 = 10. Thus oxygen‑18 contains ten neutrons, while the stable isotope oxygen‑16 has eight neutrons. This simple arithmetic underpins everything from nuclear reactors to isotope‑labeling experiments.
Beyond Counting: Why the Neutron Number Matters
1. Isotopic Stability and Decay
The neutron-to-proton ratio controls an atom’s stability. For light elements such as oxygen, a ratio close to one (e.g.That's why , 8 neutrons for 8 protons) yields a stable nucleus. When the ratio deviates—too many or too few neutrons—the nucleus becomes energetically unfavorable and may undergo beta decay or other radioactive processes. Understanding this balance is crucial in fields ranging from nuclear medicine (where short‑lived isotopes are used for imaging) to nuclear waste management.
It sounds simple, but the gap is usually here It's one of those things that adds up..
2. Nuclear Reaction Pathways
In stellar nucleosynthesis, the number of neutrons determines how a nucleus captures particles or fissions. But for example, in the slow neutron capture process (s‑process), iron seeds gradually absorb neutrons, stepping up to heavier elements. A single extra neutron can open a different reaction channel, leading to a distinct isotope that may be a key tracer of stellar evolution Easy to understand, harder to ignore..
3. Material Science and Engineering
Neutron scattering techniques exploit the fact that neutrons interact with nuclei rather than electrons. Knowing the exact neutron count in a material’s constituent atoms helps interpret scattering data, revealing crystal structures, magnetic ordering, and dynamic processes at the atomic scale. In aerospace and nuclear industries, isotopic composition can affect material performance under radiation Not complicated — just consistent. Practical, not theoretical..
Practical Tips for Working with Isotopes in the Lab
| Step | Action | Why It Matters |
|---|---|---|
| 1. Plus, acquire a High‑Purity Sample | Use isotopically enriched sources when possible. | Reduces background noise and increases signal strength, especially for NMR or mass spectrometry. |
| 2. Verify Mass Spectrometry Calibration | Confirm the mass analyzer’s accuracy with known standards. | Small mass differences (e.Think about it: g. Because of that, , 17 vs. Even so, 18 amu) require high resolution to avoid misidentification. On the flip side, |
| 3. Account for Isotopic Fractionation | Correct measured ratios for mass‑dependent fractionation when interpreting environmental data. | Prevents systematic bias in paleoclimatic reconstructions. |
| 4. Handle Radioactive Isotopes with Care | Follow institutional safety protocols and regulatory guidelines. | Protects personnel and the environment from ionizing radiation exposure. |
Concluding Thoughts
The humble act of subtracting a number—Mass Number – Atomic Number—reveals a neutron count that unlocks a universe of scientific insight. From the subtle shifts in NMR spectra that decode protein folding, to the deep‑time climate signals etched in polar ice, to the precise control of nuclear reactions that power reactors and medical diagnostics, neutrons play a silent but indispensable role.
In the grand tapestry of chemistry and physics, each neutron is a thread that, when counted and understood, weaves together disciplines, technologies, and our comprehension of the natural world. Whether you’re a student just learning the basics or a seasoned researcher probing the edges of knowledge, remembering that the neutron count is more than a number will guide you to deeper, more nuanced explorations.