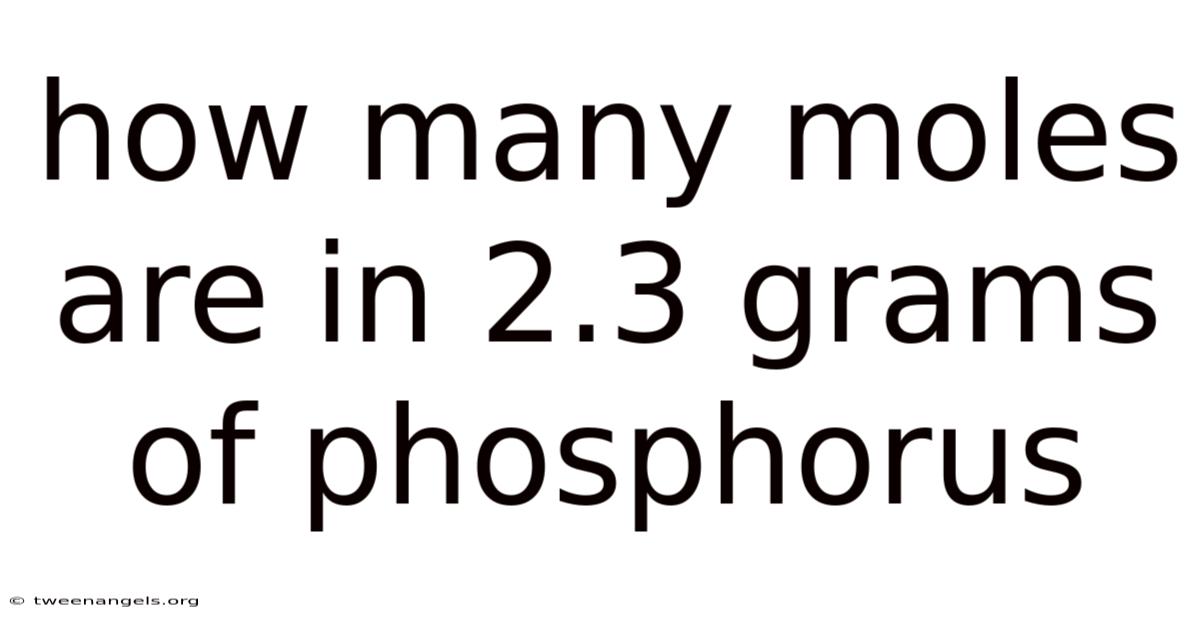Understanding the number of moles in a given mass is a fundamental concept in chemistry, especially when dealing with elements like phosphorus. When you are asked to calculate the number of moles from a specific mass, it involves a simple yet crucial formula. That's why this article will guide you through the process of determining how many moles are present in 2. 3 grams of phosphorus, ensuring clarity and precision in your calculations Most people skip this — try not to..
Phosphorus is an essential element in various biological processes and is widely used in agriculture, industry, and even in the production of fertilizers. Also, knowing the molar mass of phosphorus helps us connect the mass of a substance to the number of molecules or atoms it contains. This knowledge is vital for students, educators, and professionals who need to work with chemical calculations daily.
To begin with, let's recall the molar mass of phosphorus. Day to day, phosphorus exists in several isotopes, but the most common one in chemical calculations is phosphorus-31, which has an atomic mass of approximately 30. 97 grams per mole. On top of that, this value is crucial because it allows us to convert grams into moles easily. Understanding this helps in accurately interpreting data in lab settings or when preparing chemical equations.
You'll probably want to bookmark this section.
Now, the task at hand is to find out how many moles are in 2.3 grams of phosphorus. The process starts with the formula that relates mass, molar mass, and moles:
$ \text{Moles} = \frac{\text{Mass (g)}}{\text{Molar Mass (g/mol)}} $
In this case, the mass we are dealing with is 2.Which means 97 g/mol. Practically speaking, 3 grams**, and the molar mass of phosphorus is **30. Plugging these values into the formula gives us a straightforward calculation And that's really what it comes down to..
First, we divide the given mass by the molar mass:
$ \text{Moles of phosphorus} = \frac{2.3\ \text{g}}{30.97\ \text{g/mol}} $
Performing the division:
$ \text{Moles} \approx 0.0745\ \text{moles} $
This result tells us that there are approximately 0.This number is significant because it helps in understanding the quantity of phosphorus available for various chemical reactions or applications. Which means 0745 moles of phosphorus in 2. 3 grams. To give you an idea, in agricultural use, knowing the molar amount is essential for calculating the right dosage of fertilizers Which is the point..
One thing worth knowing that the accuracy of this calculation depends on the precision of the molar mass used. Here's the thing — 97 g/mol** is the widely accepted value for phosphorus in standard chemical references. Practically speaking, different sources may provide slightly varying values, but **30. This consistency is what makes our calculation reliable and useful Easy to understand, harder to ignore..
When working with such calculations, it's also helpful to break down the process into smaller steps. First, we identify the mass we are interested in—2.In practice, 3 grams. Then, we look up the molar mass of phosphorus, which is 30.Practically speaking, 97 g/mol. Next, we perform the division to find the number of moles. This step is crucial because it transforms the mass into a more manageable unit, allowing us to interpret the result in a meaningful way.
Another point to consider is the significance of this value in real-world applications. Because of that, for example, if you are working on a chemistry project or a lab experiment involving phosphorus, knowing that there are about 0. 0745 moles in a given mass helps in estimating the amount of product you might obtain. This is particularly useful when dealing with stoichiometric calculations, where the ratio of reactants to products is essential That alone is useful..
Also worth noting, understanding the concept of moles is not just about numbers; it's about grasping the relationship between mass and the number of particles. This connection between mass and particles is what makes chemistry so powerful and precise. Each mole of phosphorus contains a specific number of atoms, which is defined by Avogadro's number. By mastering this concept, you gain a deeper appreciation for the science behind everyday phenomena.
In addition to the calculation, it's worth exploring how this number relates to other chemical concepts. Here's a good example: if you were to convert this molar amount into grams, you could multiply it by the molar mass again. That said, in this case, we are already working with moles, which is the standard unit for such calculations. This highlights the importance of unit consistency in scientific work.
Real talk — this step gets skipped all the time Small thing, real impact..
The process of calculating moles is not only about numbers but also about developing problem-solving skills. It encourages you to think critically about the relationships between different quantities in chemistry. Whether you are a student preparing for an exam or a professional in the field, mastering this skill is invaluable Not complicated — just consistent..
It is also interesting to consider the implications of this calculation in different contexts. Here's one way to look at it: if you were to weigh out 2.In practice, 3 grams of phosphorus for a specific experiment, knowing the mole count helps in determining the exact amount needed. This practical application underscores the relevance of chemistry in real-life scenarios And that's really what it comes down to..
All in all, calculating the number of moles in 2.3 grams of phosphorus is a straightforward yet important exercise. And it reinforces the understanding of molar mass and its role in chemical calculations. By following the steps clearly and applying the correct formula, you can confidently determine the molar quantity in any given mass. This knowledge not only enhances your academic performance but also prepares you for more complex chemical challenges in the future.
Remember, every calculation is a step toward deeper understanding. Whether you are learning for the first time or refining your skills, this exercise is a great way to strengthen your grasp of fundamental concepts. With practice, you’ll find that these calculations become second nature, boosting your confidence in the lab or classroom. Let’s dive deeper into the details and ensure you have a solid grasp of this essential topic.
Now, let's apply this knowledge to the specific case at hand. To calculate the number of moles in 2.3 grams of phosphorus, we use the fundamental formula:
Number of moles = Mass (g) ÷ Molar mass (g/mol)
Phosphorus has a molar mass of approximately 31 grams per mole. Therefore:
Moles of phosphorus = 2.3 g ÷ 31 g/mol ≈ 0.074 moles
This result tells us that 2.074 moles, or about 4.3 grams of phosphorus contains approximately 0.45 × 10²² individual phosphorus atoms And that's really what it comes down to. Still holds up..
It's worth noting that phosphorus commonly exists as a tetratomic molecule (P₄) in its most stable molecular form. On top of that, this molecular structure is particularly relevant when considering reactions involving white phosphorus, which is highly reactive and characteristic of this molecular arrangement. Understanding whether you're working with atomic phosphorus or molecular phosphorus can affect certain calculations, particularly those involving gas laws or stoichiometry in specific reactions.
When performing these calculations, several common pitfalls should be avoided. Second, pay attention to significant figures—in this case, 2.On the flip side, first, always ensure you're using the correct molar mass for the specific form of the element you're working with. That's why 3 grams suggests two significant figures, so your answer should reflect this precision. Third, double-check your units; mixing grams with milligrams or using incorrect molar mass values will lead to erroneous results.
This calculation serves as a foundation for more complex stoichiometric problems. Take this case: if you were determining how many atoms would be present, you would multiply the number of moles by Avogadro's number (6.Now, 022 × 10²³). Similarly, if you needed to find how many grams of phosphorus would be required to react completely with a given amount of another substance, this mole calculation would be your starting point.
The beauty of this approach lies in its universal applicability. Even so, whether you're working with phosphorus, iron, oxygen, or any other element, the same principle applies. This consistency is what makes chemistry such a powerful and predictable science Most people skip this — try not to..
As you continue your studies, you'll find that mole calculations form the backbone of virtually every chemical reaction you'll encounter. From simple acid-base neutralizations to complex organic synthesis, the ability to accurately convert between mass and moles is an indispensable skill.
Simply put, the process of determining moles from mass is a fundamental chemical calculation that connects the macroscopic world of measurable quantities with the microscopic world of atoms and molecules. Practically speaking, 3 grams of phosphorus, this translates to approximately 0. In practice, 074 moles—a small quantity that nonetheless represents trillions upon trillions of atoms. For 2.This remarkable connection between the tangible and the infinitesimal is at the very heart of chemistry, and mastering it opens the door to understanding the layered dance of particles that governs all chemical phenomena.