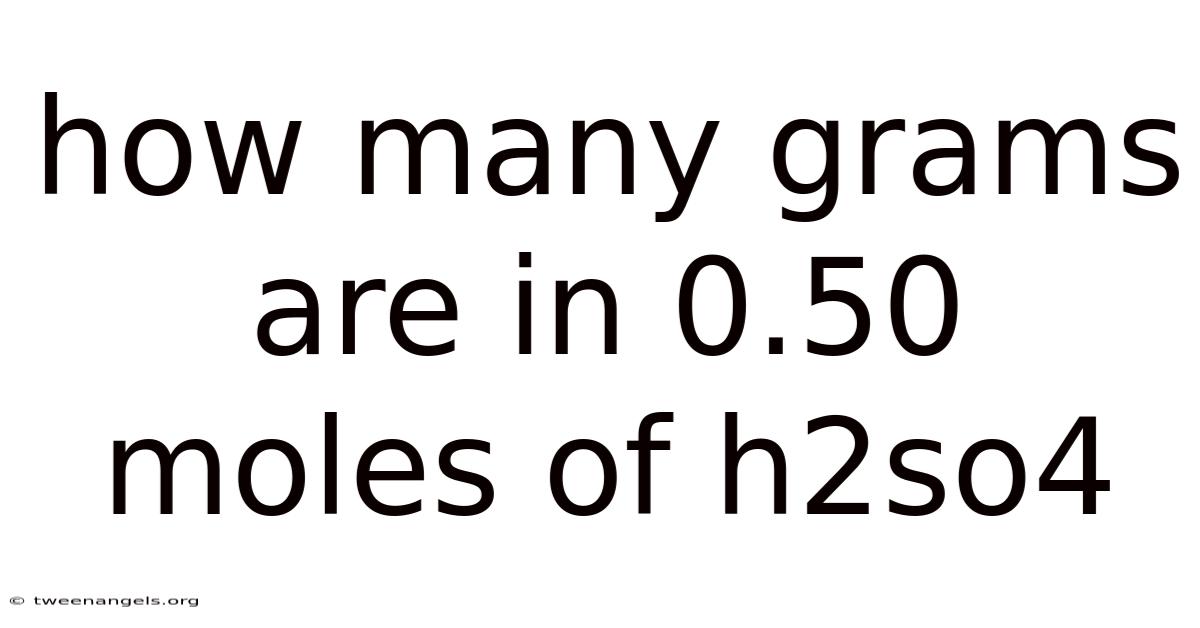Understanding molar conversions is fundamental in chemistry, serving as the bridge between abstract concepts and tangible measurements. When dealing with chemical reactions and stoichiometry, precision becomes key, enabling scientists and educators to accurately translate theoretical quantities into practical applications. Still, this article breaks down the essential process of converting moles to grams for compounds such as sulfuric acid (H₂SO₄), providing clarity on how foundational principles underpin everyday laboratory practices and industrial processes. Whether calculating reagents for a reaction or determining the quantity needed for a project, mastering these calculations ensures accuracy and efficiency across disciplines. The process involves grasping the relationship between moles, molecular weight, and mass, transforming theoretical knowledge into actionable results that resonate universally. Such foundational understanding not only simplifies complex tasks but also fosters confidence among learners and professionals alike, making it indispensable in both academic settings and professional environments. The significance extends beyond mere computation; it underpins safety protocols, environmental management, and technological advancements, reinforcing its role as a cornerstone in scientific discourse. Such precision underscores the interconnectedness of chemistry with real-world applications, where even minor inaccuracies can lead to cascading consequences. So by mastering these principles, individuals gain the tools necessary to work through the intricacies of chemical systems effectively, ensuring their contributions align with both scientific rigor and practical demands. Such knowledge empowers practitioners to troubleshoot issues, optimize processes, and innovate within their domains, thereby enhancing overall productivity and outcomes. Consider this: the process demands careful attention to detail, yet it rewards those who approach it methodically, reinforcing discipline and expertise. In practice, through this lens, we explore how molar conversions form the basis for countless calculations that shape the landscape of modern science and industry alike. The journey begins with recognizing the core concepts that govern these transformations, setting the stage for deeper exploration into their application. Such foundational insights are not merely academic exercises but practical necessities that permeate various facets of life, making them a vital component of scientific literacy. Here, we embark on a journey to unravel the mechanics behind converting moles into grams, a task that demands both technical proficiency and meticulous attention to detail. The interplay between theoretical understanding and practical application becomes evident here, highlighting why this conversion remains a critical skill across fields ranging from academia to manufacturing. So through this exploration, we aim to illuminate how such calculations serve as a universal language, facilitating seamless communication and collaboration among participants involved in chemical workflows. The process itself, though straightforward at its core, requires a disciplined approach that prioritizes accuracy and consistency, ensuring that results remain reliable and trustworthy.
The interplay between precision and adaptability continues to define its enduring relevance, bridging gaps where ambiguity once posed challenges. This leads to as disciplines evolve, so too must our grasp of interconnected systems, demanding constant refinement yet steadfast commitment. Such synergy not only elevates individual capabilities but also strengthens collective outcomes, ensuring sustainability and adaptability in an ever-changing world. The bottom line: mastery here transcends technical mastery, becoming a catalyst for progress that resonates far beyond immediate applications, cementing its role as a guiding force in shaping the future. A commitment to such understanding thus stands as a cornerstone, nurturing growth and fostering harmony across domains.
The journey into molar conversions deepens as we confront the practical nuances that arise in complex scenarios. Similarly, in environmental chemistry, quantifying pollutant concentrations often involves converting moles of a contaminant (detected via titration or spectroscopy) into grams per liter to assess ecological impact and guide remediation strategies. Consider the synthesis of a novel polymer in materials science; accurately calculating the mass of monomers required per batch hinges entirely on precise mole-to-gram conversions, directly influencing material properties like tensile strength and thermal stability. These applications underscore the conversion's role as more than a mere arithmetic step; it is the critical link between theoretical stoichiometry and tangible outcomes.
Not the most exciting part, but easily the most useful Not complicated — just consistent..
What's more, the adaptability of mole-to-gram conversions becomes essential when dealing with non-ideal conditions. Take this case: in pharmaceutical manufacturing, calculating the mass of an active pharmaceutical ingredient (API) needed for a specific formulation must account for hydration states (e.Now, g. , moles of anhydrous vs. And hydrated compound) and potential impurities, demanding meticulous attention to molar mass precision. This adaptability extends to analytical chemistry, where converting moles of analyte detected into grams allows for the calibration of instruments and the validation of experimental results against known standards, ensuring data integrity across laboratories Worth keeping that in mind..
When all is said and done, the mastery of converting moles to grams is not an isolated skill but a foundational competency that permeates the scientific and industrial landscape. It empowers researchers to design dependable experiments, engineers to optimize processes with confidence, and policymakers to interpret data with clarity. This fluency in the quantitative language of chemistry transcends disciplinary boundaries, fostering collaboration and enabling the translation of molecular-scale understanding into macro-scale solutions. Here's the thing — as we work through an increasingly complex world, where chemical literacy underpins innovation in sustainability, medicine, and technology, the ability to easily manage molar conversions remains an indispensable tool, bridging the microscopic realm of atoms and molecules with the tangible realities of human progress. It is this precise, adaptable, and universally applicable understanding that truly solidifies chemistry's role as the central science driving our collective future.
In an era defined by rapid technological advancement, mastering molar conversions remains a cornerstone, enabling precise control over material development and sustainable practices. Now, this precision not only enhances scientific accuracy but also supports global efforts to address pressing challenges, proving its enduring relevance. Thus, as chemistry evolves alongside society, the ability to handle these conversions remains vital, ensuring that the foundational principles continue to underpin progress.
Conclusion: Such expertise acts as a bridge, connecting microscopic intricacies to macroscopic impact, fostering innovation and resilience across disciplines. It remains a testament to chemistry's important role in shaping the world, urging careful stewardship as demands grow ever more complex Surprisingly effective..