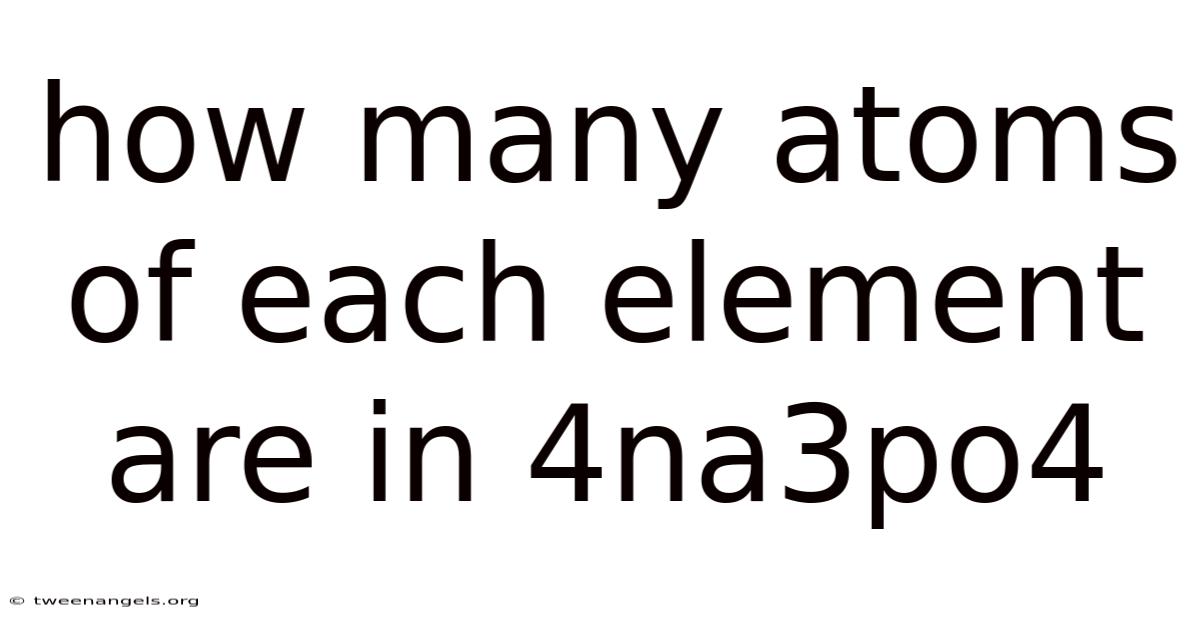The involved dance of atoms within molecules forms the very foundation of the physical universe, governing everything from the stability of matter to the behavior of celestial bodies. The process involves translating chemical formulas into atomic quantities, a task that requires both mathematical precision and a deep comprehension of atomic theory. While many people intuitively grasp the composition of substances like water or sodium chloride, the precise calculation required for more complex formulas such as 4Na₃PO₄ presents a challenge that demands careful attention to detail. That's why such knowledge serves as a cornerstone in fields ranging from chemistry laboratories to materials engineering, where the manipulation of atomic arrangements directly impacts performance, safety, and efficiency. Within this framework lies a fundamental yet often overlooked concept: determining the precise number of atoms composing a specific chemical compound. On top of that, this endeavor is not merely an academic exercise but a practical necessity across scientific research, industrial applications, and even everyday problem-solving. Through this process, one uncovers the silent architects behind the material universe itself, revealing how even the simplest elements combine in specific ratios to form the building blocks of matter as we know it. On the flip side, understanding how atoms contribute to the overall structure of compounds allows scientists to predict properties, design materials, and innovate technologies that shape our world. The challenge lies not only in accuracy but also in ensuring that the results are interpretable and applicable to real-world scenarios, making the task both demanding and profoundly rewarding And that's really what it comes down to. No workaround needed..
Understanding Atomic Composition in Chemical Formulations
To grasp the intricacies of 4Na₃PO₄, one must first dissect its molecular structure and identify the constituent elements. At its core, sodium phosphate (often abbreviated as Na₃PO₄) consists of three sodium ions (Na⁺) bonded to one phosphate group (PO₄³⁻). When scaled up to four units of this compound, the arrangement becomes clearer: four sodium ions, twelve phosphate ions, and sixteen oxygen atoms per formula unit. This structural clarity is crucial because it dictates how atoms interact within the compound. Sodium, being a Group 1 metal, readily donates one electron per atom, while phosphate, a polyatomic ion, carries a negative charge stabilized by shared electrons. Understanding these roles helps contextualize why precise atomic counts matter—each sodium contributes to the compound’s overall charge balance, and each phosphate unit ensures stability through its high valency. Worth adding, recognizing the atomic proportions allows for the calculation of total atoms within the entire compound. Here's one way to look at it: if one molecule of 4Na₃PO₄ contains 4×(1 Na + 3 P + 4 O) atoms, the math reveals a total of 16 sodium, 48 phosphorus, and 64 oxygen atoms. Such calculations are not trivial but form the basis for predicting how these elements influence the compound’s physical and chemical behaviors. The precision required here underscores the importance of foundational knowledge in chemistry, where even minor errors can lead to significant misinterpretations.
Breaking Down the Mathematical Framework
The mathematical foundation underpinning this calculation rests on understanding molar ratios and stoichiometric principles. At the heart of chemistry lies the concept of molar mass, which represents the average mass of a molecule or ion in grams per mole. For 4Na₃PO₄, determining its molar mass is essential first step. Sodium (Na) has a molar mass of approximately 23 g/mol, phosphorus (P
Continuing the mathematical breakdown, phosphorus (P) contributes approximately 31 g/mol, while oxygen (O) is about 16 g/mol. Calculating the molar mass of a single formula unit of Na₃PO₄ involves summing the contributions:
(3 × Na) + (1 × P) + (4 × O) = (3 × 23) + (1 × 31) + (4 × 16) = 69 + 31 + 64 = 164 g/mol.
That said, the compound under examination is 4Na₃PO₄, signifying four discrete formula units. That's why, its total molar mass is:
4 × 164 g/mol = 656 g/mol Not complicated — just consistent..
This step is foundational, as molar mass serves as the bridge between the macroscopic scale (grams, measurable in labs) and the atomic scale (atoms, molecules). It allows chemists to quantify substances precisely, enabling reactions to be balanced accurately and solutions to be formulated with exact concentrations Simple, but easy to overlook..
Counting Atoms: Precision in Practice
With the molar mass established, the next logical step is enumerating the individual atoms within 4Na₃PO₄. Each formula unit (Na₃PO₄) contains:
- 3 Sodium (Na) atoms
- 1 Phosphorus (P) atom
- 4 Oxygen (O) atoms
Scaling this to four formula units (4Na₃PO₄):
- Sodium (Na): 4 formula units × 3 Na/unit = 12 Na atoms
- Phosphorus (P): 4 formula units × 1 P/unit = 4 P atoms
- Oxygen (O): 4 formula units × 4 O/unit = 16 O atoms
This meticulous count reveals the true atomic composition: 12 Sodium, 4 Phosphorus, and 16 Oxygen atoms. This clarity is indispensable. It explains why 4Na₃PO₄, despite appearing as a single entity, is fundamentally an aggregate of these specific elements in fixed proportions. This leads to understanding this composition is critical for predicting reactivity, solubility, and interactions with other substances. To give you an idea, the high oxygen content relative to phosphorus influences its behavior as a water softener or detergent builder, where oxygen atoms make easier ion exchange Small thing, real impact. No workaround needed..
Conclusion
The analysis of 4Na₃PO₄ exemplifies the profound importance of atomic precision in chemistry. By dissecting its structure, calculating its molar mass, and enumerating its constituent atoms, we move beyond symbols on a page to grasp the tangible reality of matter. Each number and ratio is a testament to chemistry's foundational principles: conservation of mass, definite proportions, and the predictable behavior of elements. This knowledge is not merely academic; it underpins advancements in fields ranging from industrial manufacturing and environmental remediation to pharmaceuticals and nanotechnology. The ability to accurately interpret and manipulate atomic compositions empowers scientists and engineers to design materials with tailored properties, develop sustainable processes, and solve complex global challenges. The bottom line: the seemingly simple act of counting atoms in a compound like 4Na₃PO₄ unlocks the door to understanding and shaping the material world, demonstrating that the smallest units hold the greatest significance in the grand tapestry of chemical science.
Practical Calculations: From Grams to Moles and Back
Having established that 4 Na₃PO₄ contains 12 Na, 4 P, and 16 O atoms, the next step for any laboratory or production environment is converting that information into usable quantities. Suppose a formulation requires 0.Worth adding: 250 mol of Na₃PO₄. Because one mole of Na₃PO₄ corresponds to 163 The details matter here..
[ 0.250\ \text{mol} \times 163.94\ \frac{\text{g}}{\text{mol}} = 40.
If the recipe calls for exactly four formula units, the mass is simply four times the molar mass divided by Avogadro’s number:
[ \frac{4 \times 163.94\ \text{g}}{6.022 \times 10^{23}} \approx 1.
While this number is far too small for practical work, it illustrates the continuity between the macroscopic and microscopic worlds. 25 × 10²⁵** oxygen atoms. Also, 13 × 10²⁴** phosphorus atoms, and **3. In industrial settings, the same principles are scaled up: a 1‑ton batch of Na₃PO₄ corresponds to roughly 6.44 × 10²⁵ sodium atoms, 8.In real terms, 10 × 10³ mol, which in turn contains **2. These astronomical figures are routinely handled by process engineers using software that tracks mole balances rather than individual atoms.
Stoichiometric Implications in Reaction Design
When Na₃PO₄ participates in a chemical reaction, the atomic count directly informs the stoichiometric coefficients. Consider the precipitation of calcium phosphate:
[ 3\ \text{Na}_3\text{PO}_4\ (aq) + 3\ \text{CaCl}_2\ (aq) \rightarrow \text{Ca}_3(\text{PO}_4)_2\ (s) + 6\ \text{NaCl}\ (aq) ]
Each mole of Na₃PO₄ supplies one mole of PO₄³⁻, which contains four oxygen atoms. By tracking the oxygen count, chemists can predict the lattice structure of the precipitate and anticipate its solubility under varying pH conditions. The four oxygen atoms are essential for forming the phosphate tetrahedron that ultimately binds three calcium ions. Also worth noting, the sodium ions released serve as counter‑cations, influencing the ionic strength of the solution—a factor that can shift equilibrium positions and affect product yield Not complicated — just consistent..
Safety, Handling, and Environmental Considerations
Na₃PO₄ is classified as a low‑hazard inorganic salt, yet its high alkalinity (pH ≈ 12 in aqueous solution) demands careful handling. That's why the abundance of oxygen atoms contributes to its strong affinity for metal cations, which can lead to rapid complexation and, in some cases, corrosive behavior toward aluminum or zinc surfaces. Personal protective equipment (gloves, goggles, and lab coats) mitigates direct contact, while engineering controls such as fume hoods prevent aerosol exposure.
From an environmental standpoint, the phosphate component can become a double‑edged sword. That said, excess phosphates discharged into natural water bodies can trigger eutrophication, fostering algal blooms that deplete dissolved oxygen. On the flip side, in wastewater treatment, phosphate salts are employed to precipitate heavy metals, leveraging the same oxygen‑rich phosphate groups that bind metal ions. Understanding the precise atomic composition of Na₃PO₄ therefore informs both its beneficial applications and the design of mitigation strategies—such as dosing calculations that avoid over‑addition That's the whole idea..
Real‑World Applications Beyond the Laboratory
-
Detergent Builders
The high oxygen content of the PO₄³⁻ ion enhances its ability to sequester calcium and magnesium ions, preventing soap scum formation. Modern “phosphate‑free” formulations often replace Na₃PO₄ with zeolites, but the legacy of Na₃PO₄ in industrial cleaning underscores its historical importance Turns out it matters.. -
Food Additive (E‑341)
Sodium phosphate acts as an emulsifier, pH regulator, and leavening agent in processed foods. The precise molar ratios of Na, P, and O dictate its buffering capacity, which is critical for maintaining product stability over shelf life Easy to understand, harder to ignore. But it adds up.. -
Water Softening
In municipal water treatment, Na₃PO₄ is dosed to precipitate hardness‑causing ions as insoluble phosphates. The stoichiometry of the precipitation reaction hinges on the availability of four oxygen atoms per phosphate, which coordinate with divalent cations to form stable mineral phases That's the part that actually makes a difference.. -
Biomedical Research
Phosphate buffers, derived from Na₃
Conclusion
To wrap this up, the chemical composition of Na₃PO₄, specifically its high oxygen content, is key here in its various applications. From laboratory settings to industrial and consumer products, the unique properties of sodium phosphate make it an essential component. Its ability to sequester metal ions, regulate pH, and form stable mineral phases has led to its widespread use in detergent builders, food additives, water softening, and biomedical research. That said, its potential environmental impact, particularly with regard to eutrophication, highlights the importance of responsible handling and dosing calculations. Consider this: as research continues to uncover new applications for Na₃PO₄, a deep understanding of its atomic composition and resulting properties will remain essential for optimizing its benefits while minimizing its risks. By acknowledging the layered relationships between the sodium, phosphorus, and oxygen atoms in Na₃PO₄, scientists and industries can harness its full potential while promoting a more sustainable future Most people skip this — try not to. Surprisingly effective..