How Many Atoms Are In 15.6 G Of Silicon
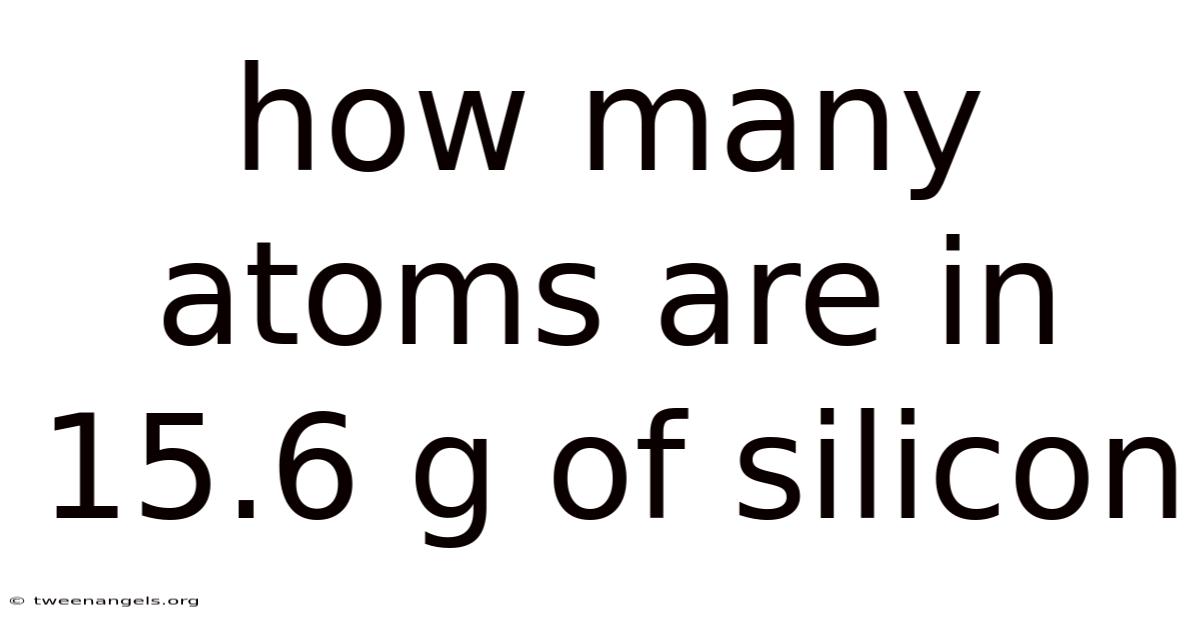
How many atoms are in 15.6 g of silicon? This question lies at the heart of stoichiometry and solid‑state chemistry, and answering it provides a clear window into the microscopic world of elements. In this article we will walk through the exact calculation, explain the underlying scientific principles, and address common follow‑up questions that arise when converting mass to particle number. By the end, you will not only have the numerical answer but also a solid grasp of the method that can be applied to any similar problem involving elemental mass and Avogadro’s number.
Introduction
Silicon (Si) is a cornerstone material in modern electronics, optics, and photovoltaics. When chemists or engineers need to know how many individual silicon atoms are present in a given macroscopic mass, they turn to the concept of the mole and Avogadro’s constant. The phrase how many atoms are in 15.6 g of silicon encapsulates a straightforward yet powerful transformation: from a measurable mass to an astronomically large count of discrete particles. Understanding this conversion is essential for tasks ranging from material budgeting in semiconductor fabrication to estimating the number of dopant atoms required for device design. The following sections break down the process step by step, provide the scientific context, and answer the most frequently asked questions.
StepsTo determine the number of silicon atoms in 15.6 g, follow these systematic steps:
-
Identify the molar mass of silicon.
The atomic weight of silicon is approximately 28.085 g mol⁻¹. This value represents the mass of one mole of silicon atoms. -
Calculate the number of moles present in the sample.
Use the formula [ \text{moles} = \frac{\text{mass}}{\text{molar mass}} = \frac{15.6\ \text{g}}{28.085\ \text{g mol}^{-1}} ]
This yields about 0.555 mol of silicon. -
Convert moles to number of atoms using Avogadro’s number.
Avogadro’s constant, (N_A), is 6.022 × 10²³ atoms mol⁻¹. Multiply the mole value by this constant:
[ \text{atoms} = 0.555\ \text{mol} \times 6.022 × 10^{23}\ \text{atoms mol}^{-1} ]
The result is roughly 3.34 × 10²³ atoms. -
Express the answer with appropriate significant figures.
Since the original mass (15.6 g) has three significant figures, round the final count to three significant figures: 3.34 × 10²³ atoms.
These steps illustrate a universal pathway: mass → moles → particles. The same approach works for any element or compound, provided you have the correct molar mass and Avogadro’s number.
Scientific Explanation### The Mole Concept
The mole is a fundamental unit in chemistry that bridges the macroscopic world (grams, liters) and the microscopic world (atoms, molecules). One mole of any substance contains exactly (N_A) entities, a number so large that it is inconvenient to write out fully but essential for quantitative chemistry. The International System of Units (SI) defines the mole in terms of the number of elementary entities, making it a stable reference point.
Molar Mass and Atomic WeightThe molar mass of an element is numerically equal to its standard atomic weight expressed in grams per mole. For silicon, the atomic weight of 28.085 u translates directly to a molar mass of 28.085 g mol⁻¹. This equivalence arises because the atomic mass unit (u) is defined such that one mole of atoms with a mass of 1 u has a mass of 1 g.
Avogadro’s Constant
Avogadro’s constant, (N_A = 6.022 × 10^{23}), is not a measured quantity but a defined constant. It was chosen to ensure that the molar mass of carbon‑12 in grams is exactly 12 g, thereby linking the atomic mass scale to the macroscopic mass scale. This constant allows chemists to count particles indirectly through mass measurements.
Significant Figures and Uncertainty
When performing calculations, the number of significant figures in the final answer should reflect the least precise measurement. In our case,
...the mass measurement (15.6 g) dictates the precision of our final answer. Reporting 3.34 × 10²³ atoms correctly reflects this limitation. In laboratory practice, uncertainties in mass, purity, or molar mass values propagate through such calculations, making the careful application of significant figures a simple yet vital tool for honest communication of precision.
Broader Utility and Interconnectivity
This mass-moles-particles framework is not an isolated exercise but the quantitative backbone of chemistry. It underpins:
- Stoichiometry: Predicting yields and reactant requirements in chemical reactions.
- Solution Chemistry: Relating molarity (mol/L) to the number of solute particles.
- Analytical Techniques: From gravimetric analysis to spectroscopy, all ultimately connect a measured signal to a count of atoms or molecules.
- Materials Science: Determining atomic densities, dopant concentrations, or nanoparticle compositions.
The elegance of this system lies in its universality. Whether dealing with elemental silicon, a complex pharmaceutical molecule, or a gaseous mixture, the same logical steps—using the appropriate molar mass and Avogadro’s constant—translate a measurable mass into an exact count of fundamental entities. This conversion is what allows chemistry to move from qualitative observations to precise, reproducible, and scalable science.
Conclusion
The calculation of atoms from a given mass is a fundamental demonstration of the mole concept’s power. By systematically applying molar mass and Avogadro’s constant, we bridge the tangible scale of the laboratory with the atomic scale of matter. This process, governed by the rules of significant figures, ensures that our quantitative descriptions are both mathematically sound and appropriately precise. Mastering this pathway is essential for any chemist, as it forms the indispensable link between the weights we measure and the particles that define the material world. It is the cornerstone upon which all quantitative chemical understanding is built.
Beyond the Basics: Temperature and Pressure Considerations
While the fundamental relationship remains constant, real-world measurements often introduce complexities. Temperature and pressure significantly impact molar mass, particularly when dealing with gases. The ideal gas law (PV = nRT) provides a crucial link: knowing the volume (V), pressure (P), temperature (T), and the number of moles (n), we can calculate the molar mass (M) of a gas: M = (PT)/RT. This equation highlights the importance of accurately determining these conditions alongside the mass measurement. Furthermore, deviations from ideal behavior, especially at high pressures or with non-ideal gases, necessitate the use of more sophisticated equations of state.
Error Propagation and Systemic Errors
It’s crucial to recognize that errors aren’t always random. Systemic errors – consistent biases in measurements – can dramatically affect the final result. For instance, a consistently inaccurate balance will lead to an overestimation or underestimation of mass. Similarly, contamination of a sample can skew the molar mass value. Understanding and mitigating these sources of error is paramount to reliable results. Error propagation techniques, often involving the square root of the sum of squares of individual uncertainties, allow chemists to estimate the overall uncertainty in their calculated particle count.
Expanding the Scope: Isotopes and Elemental Composition
The concept extends beyond single elements to encompass mixtures and isotopes. When dealing with a compound containing multiple elements, the molar mass must be calculated as a weighted average based on the element’s atomic mass and its proportion in the compound. Similarly, isotopes of an element have slightly different masses, and these variations must be accounted for when determining the number of atoms. Spectroscopic techniques, like mass spectrometry, provide direct measurements of isotopic ratios, offering a powerful tool for elemental analysis and compound identification.
Conclusion
The seemingly simple calculation of atoms from mass represents a profound and elegantly constructed framework within chemistry. From the foundational principles of the mole to the consideration of temperature, pressure, and isotopic variations, this process underscores the interconnectedness of measurement, calculation, and the fundamental nature of matter. By diligently applying the rules of significant figures, acknowledging potential errors, and expanding our understanding to encompass complex mixtures and isotopic compositions, chemists can confidently translate the macroscopic world of weights and volumes into the microscopic realm of atoms and molecules – a cornerstone of scientific progress and technological innovation.
Latest Posts
Latest Posts
-
The Elongation Of The Leading Strand During Dna Synthesis
Mar 24, 2026
-
What Is The Difference Between A Diploid And Haploid Cell
Mar 24, 2026
-
Your Goal In Framing A Business Message Is To
Mar 24, 2026
-
Which Disorder Would The Nurse Classify As Neurodevelopmental
Mar 24, 2026
-
Find The Domain Of The Rational Expression
Mar 24, 2026