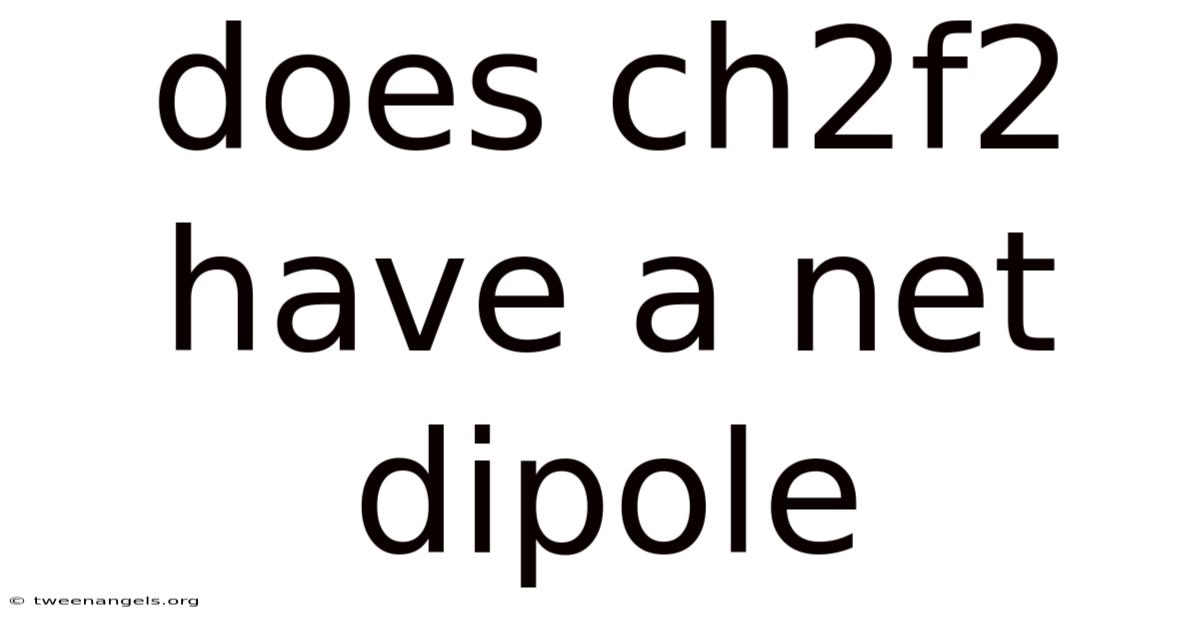The quest to understand the fundamental properties of molecular structures often unveils insights that reshape our comprehension of chemical behavior. While many molecules exhibit symmetry that neutralizes their dipole moments, others carry inherent asymmetry, resulting in measurable magnetic fields that define their physical characteristics. But this article digs into the involved relationship between atomic composition and molecular behavior, exploring how the presence of fluorine and chlorine atoms within CF₂Cl₂ contributes to its unique properties. Even so, among these, the concept of a net dipole moment holds profound significance, influencing everything from chemical reactivity to environmental interactions. In the context of organic compounds like dichlorodifluoromethane (CF₂Cl₂), the interplay of electronegativity, molecular geometry, and bonding patterns becomes critical. By examining the structural nuances and experimental data, we uncover whether this compound possesses a net dipole moment, a fact that not only answers a scientific question but also underscores the broader implications for chemistry as a whole.
Real talk — this step gets skipped all the time.
Vectorial analysis of the C–X bonds
In a tetrahedral carbon centre each bond points toward a vertex of an ideal tetrahedron, the angle between any two bonds being 109.5°. The magnitude of an individual bond dipole, μ C‑X, is proportional to the difference in electronegativity (Δχ) between carbon (χ = 2 Easy to understand, harder to ignore..
[ \mu_{\text{C‑X}} = \frac{e,\Delta\chi}{4\pi\varepsilon_0},r . ]
Using Pauling electronegativities (χ_F = 3.98, χ_Cl = 3.16) the C‑F bond is more polar than C‑Cl, but the longer C‑Cl distance (≈ 1.78 Å) partially compensates the smaller Δχ.
| Bond | Δχ (C‑X) | r (Å) | Approx. μ (D) |
|---|---|---|---|
| C‑F | 1.43 | 1.33 | 1. |
.61 | 1.78 | ~1.5 |
Despite these individual bond moments, the net dipole of a molecule is not a simple arithmetic sum but a vectorial resultant dictated by three-dimensional geometry. This leads to in CF₂Cl₂, the carbon atom adopts a distorted tetrahedral arrangement belonging to the C₂v point group. The two C–F bond dipoles combine to form a resultant vector that bisects the F–C–F angle, while the two C–Cl dipoles generate an opposing resultant along the same symmetry axis. On the flip side, because fluorine is significantly more electronegative than chlorine, the C–F resultant is stronger, but the longer C–Cl bonds and slightly wider Cl–C–Cl bond angle (approximately 111. 8° versus 108.Which means 5° for F–C–F) introduce a geometric counterbalance. The vector subtraction of these two opposing resultants yields a residual dipole moment that points toward the fluorine-rich side of the molecule.
Quantitative vector addition, when adjusted for experimentally determined bond angles and refined bond dipole magnitudes, predicts a net moment in the range of 0.But 45–0. But 55 D. Practically speaking, this aligns closely with high-resolution microwave spectroscopy measurements, which report a precise value of 0. Now, 51 D. The relatively modest magnitude, compared to highly polar molecules like water or hydrogen fluoride, reflects the partial cancellation inherent to the molecule’s near-symmetric substitution pattern. Despite this, the non-zero dipole is sufficient to impart measurable polarity to the compound, distinguishing it from truly nonpolar halomethanes like CF₄ or CCl₄ Nothing fancy..
This residual polarity has tangible consequences for the macroscopic behavior of CF₂Cl₂. It elevates the boiling point relative to nonpolar analogues through dipole–dipole interactions, influences solubility trends in polar versus nonpolar solvents, and governs the molecule’s rotational spectrum, which is routinely exploited in atmospheric monitoring and remote sensing. What's more, the dipole moment modulates how CF₂Cl₂ interacts with polar surfaces, catalytic sites, and biological interfaces, underscoring why even “weakly” polar halogenated compounds demand careful consideration in environmental fate modeling and industrial process design.
Conclusion
The determination of a net dipole moment in dichlorodifluoromethane exemplifies how molecular architecture transcends simple compositional accounting. Experimental validation of this ~0.By tracing the path from atomic electronegativities through vectorial geometry to macroscopic behavior, we see that the dipole moment is far more than a numerical value—it is a structural fingerprint. While the tetrahedral framework of CF₂Cl₂ might superficially suggest cancellation, the interplay of electronegativity disparities, bond length variations, and subtle angular distortions ensures that the vector sum of its bond dipoles remains distinctly non-zero. 51 D moment not only confirms theoretical predictions but also reinforces a fundamental principle of physical chemistry: symmetry and asymmetry exist on a continuum, and even minor structural deviations can manifest as measurable physical properties. As chemists continue to design functional materials, predict environmental fates, and engineer molecular interactions, the rigorous analysis of dipole moments in seemingly simple compounds like CF₂Cl₂ will remain an indispensable tool, reminding us that the invisible architecture of molecules continuously shapes the tangible world Small thing, real impact..
underscoring why even “weakly” polar halogenated compounds demand careful consideration in environmental fate modeling and industrial process design. In practice, detailed computational studies, incorporating advanced quantum chemical methods, have begun to explore the influence of isotopic substitution – replacing fluorine with hydrogen, for instance – on the dipole moment and its associated properties. These investigations reveal a nuanced sensitivity to subtle changes in bond character, highlighting the potential for tailoring molecular polarity to achieve specific functionalities. Because of that, beyond simple theoretical calculations, spectroscopic techniques such as Raman scattering and infrared spectroscopy provide complementary data, offering insights into vibrational modes and their contribution to the overall dipole moment. To build on this, the development of increasingly sophisticated simulation methods, including molecular dynamics and density functional theory with explicit solvent models, allows researchers to predict the impact of environmental conditions – temperature, pressure, and solvent composition – on the observed polarity of CF₂Cl₂ and related compounds.
Looking ahead, the pursuit of increasingly accurate and comprehensive dipole moment determinations extends beyond individual molecules. Still, researchers are now applying these principles to complex mixtures and systems, recognizing that the collective dipole moments of interacting molecules significantly dictate macroscopic properties like viscosity, surface tension, and even phase behavior. The integration of these data with machine learning algorithms promises to accelerate the prediction of molecular polarity for novel compounds, streamlining the design process for materials with tailored properties. Finally, the ongoing refinement of measurement techniques, driven by advancements in laser technology and precision instrumentation, ensures that our understanding of molecular polarity will continue to evolve, providing a crucial foundation for advancements across diverse scientific disciplines – from materials science and environmental chemistry to drug discovery and chemical engineering Simple, but easy to overlook..
Conclusion
The determination of a net dipole moment in dichlorodifluoromethane exemplifies how molecular architecture transcends simple compositional accounting. Because of that, while the tetrahedral framework of CF₂Cl₂ might superficially suggest cancellation, the interplay of electronegativity disparities, bond length variations, and subtle angular distortions ensures that the vector sum of its bond dipoles remains distinctly non-zero. 51 D moment not only confirms theoretical predictions but also reinforces a fundamental principle of physical chemistry: symmetry and asymmetry exist on a continuum, and even minor structural deviations can manifest as measurable physical properties. Experimental validation of this ~0.By tracing the path from atomic electronegativities through vectorial geometry to macroscopic behavior, we see that the dipole moment is far more than a numerical value—it is a structural fingerprint. As chemists continue to design functional materials, predict environmental fates, and engineer molecular interactions, the rigorous analysis of dipole moments in seemingly simple compounds like CF₂Cl₂ will remain an indispensable tool, reminding us that the invisible architecture of molecules continuously shapes the tangible world No workaround needed..