Density Of Lead In G Cm3
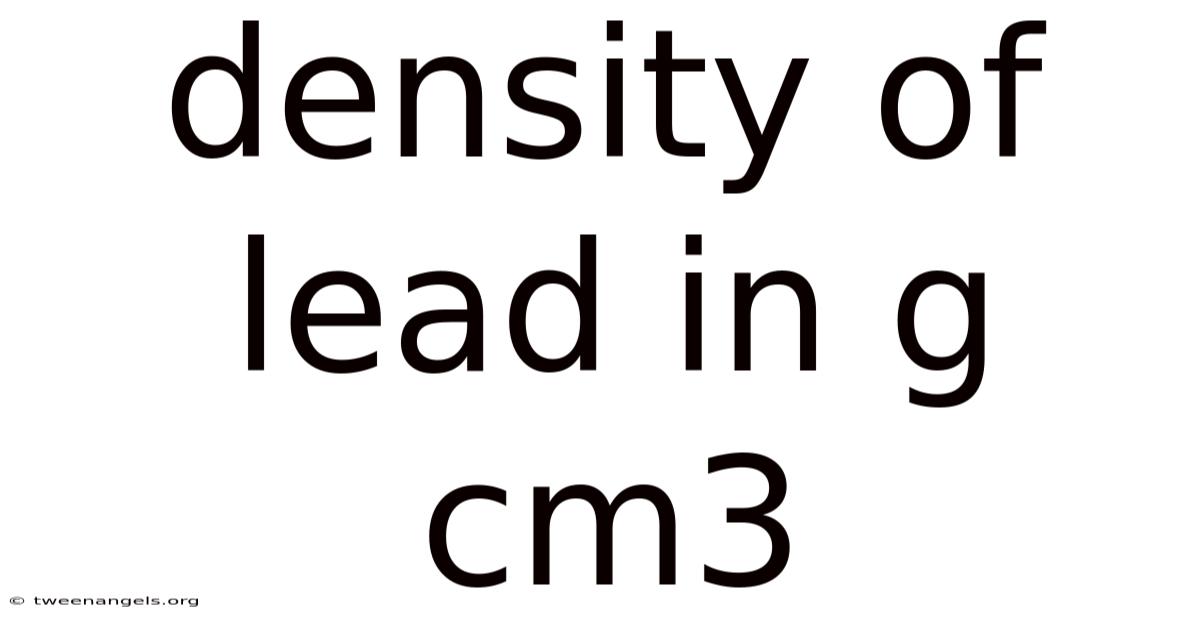
Density of Lead in g/cm³: Understanding Its Properties and Applications
The density of lead in g/cm³ is 11.34, making it one of the densest common materials on Earth. This remarkable property has made lead valuable for numerous applications throughout human history, from ancient water pipes to modern radiation shielding. Understanding the density of lead is essential for various scientific, industrial, and safety considerations, as it directly influences how this metal behaves in different environments and applications.
Understanding Density
Density is defined as the mass of a substance per unit volume, typically expressed in grams per cubic centimeter (g/cm³) for solids and liquids. The mathematical formula for density is ρ = m/V, where ρ represents density, m stands for mass, and V denotes volume. This fundamental physical property helps identify materials and predict their behavior under different conditions.
The density of a material remains relatively constant regardless of sample size, making it a characteristic property that distinguishes one substance from another. For lead, this value of 11.34 g/cm³ means that a cubic centimeter of lead weighs 11.34 grams, significantly more than most other common metals.
Lead's Density: The Numbers
Pure lead has a density of 11.34 g/cm³ at room temperature (20°C or 68°F) and standard atmospheric pressure. This value can vary slightly depending on the specific isotopic composition, temperature, and pressure conditions. When lead is alloyed with other metals, its density may decrease slightly depending on the other elements present and their proportions.
Several factors can affect the measured density of lead:
- Temperature: As temperature increases, most materials expand slightly, causing density to decrease.
- Purity: Impurities and alloying elements can alter the density.
- Crystal structure: Different allotropes of lead may have slightly different densities.
- Porosity: Cast lead with internal air pockets will have lower effective density.
Why Lead is So Dense
Lead's exceptional density stems from its atomic structure and properties. With an atomic number of 82, lead is a heavy element with a high atomic mass of 207.2 g/mol. Its atoms are relatively large and pack closely together in a face-centered cubic crystal structure, contributing to its high mass per unit volume.
The position of lead in the periodic table, in the sixth period and group 14, gives it a large atomic radius and high atomic mass. These factors combine to create the high density characteristic of lead. The stability of its electron configuration also contributes to its dense atomic packing.
Scientific Explanation of Lead's Density
From a quantum mechanical perspective, lead's density can be understood through its electron configuration. Lead has a completely filled 4f subshell and a stable 6s² electron configuration. This electron arrangement contributes to its metallic bonding characteristics and dense atomic packing.
The crystal structure of lead is face-centered cubic (FCC), which is one of the most efficient packing arrangements for atoms. In this structure, each atom has 12 nearest neighbors, allowing for maximum atomic packing efficiency. When combined with lead's high atomic mass, this results in the high observed density.
Practical Applications of Lead's Density
Lead's exceptional density makes it valuable in numerous applications:
- Radiation shielding: Lead effectively blocks X-rays, gamma rays, and other forms of radiation due to its high electron density and atomic number.
- Ballistics: The density of lead makes it ideal for ammunition, as it maintains momentum effectively when fired.
- Counterweights: In machinery and systems requiring balance, lead's high density allows for smaller, more compact counterweights.
- Soundproofing: Lead's density helps block sound transmission in acoustic applications.
- Fishing sinkers: The density of lead allows for small, heavy sinkers that help fishing lines sink quickly.
Comparison with Other Metals
When compared to other common metals, lead's density stands out:
- Gold: 19.32 g/cm³
- Osmium: 22.59 g/cm³ (densest naturally occurring element)
- Platinum: 21.45 g/cm³
- Tungsten: 19.25 g/cm³
- Uranium: 19.1 g/cm³
- Lead: 11.34 g/cm³
- Silver: 10.49 g/cm³
- Copper: 8.96 g/cm³
- Iron: 7.87 g/cm³
- Aluminum: 2.70 g/cm³
While lead is not the densest element, it is more accessible and easier to work with than many denser alternatives, making it practical for many applications.
Safety Considerations
Despite its useful properties, lead is toxic and requires careful handling. The density of lead means that even small amounts can contain significant mass, increasing the risk of exposure if not handled properly. Safety precautions include:
- Wearing protective equipment when handling lead
- Ensuring proper ventilation in work areas
- Avoiding ingestion or inhalation of lead particles
- Following proper disposal procedures for lead waste
- Monitoring blood lead levels for workers with regular exposure
Frequently Asked Questions About Lead Density
Q: Why is lead density measured in g/cm³? A: The g/cm³ unit is commonly used for density measurements of solids and liquids because it provides convenient values for everyday materials. Lead's density of 11.34 g/cm³ means a cubic centimeter weighs 11.34 grams.
Q: Does the density of lead change when it's alloyed with other metals? A: Yes, when lead is alloyed with other elements, its density typically decreases depending on the density of the alloying elements and their proportions. For example, solder (lead-tin alloy) has a lower density than pure lead.
Q: How does temperature affect lead's density? A: Like most materials, lead expands when heated, causing its density to decrease. The coefficient of thermal expansion for lead is approximately 29 × 10⁻
Latest Posts
Latest Posts
-
Which Rna Base Bonds With Guanine
Mar 25, 2026
-
Strategic Brand Management Building Measuring And Managing Brand Equity
Mar 25, 2026
-
Ccl4 Lewis Structure Polar Or Nonpolar
Mar 25, 2026
-
Health Safety And Nutrition For The Young Child
Mar 25, 2026
-
Interest Rates Are Expressed As A Percentage Of
Mar 25, 2026