Cis 1 Tert Butyl 4 Methylcyclohexane
tweenangels
Mar 15, 2026 · 7 min read
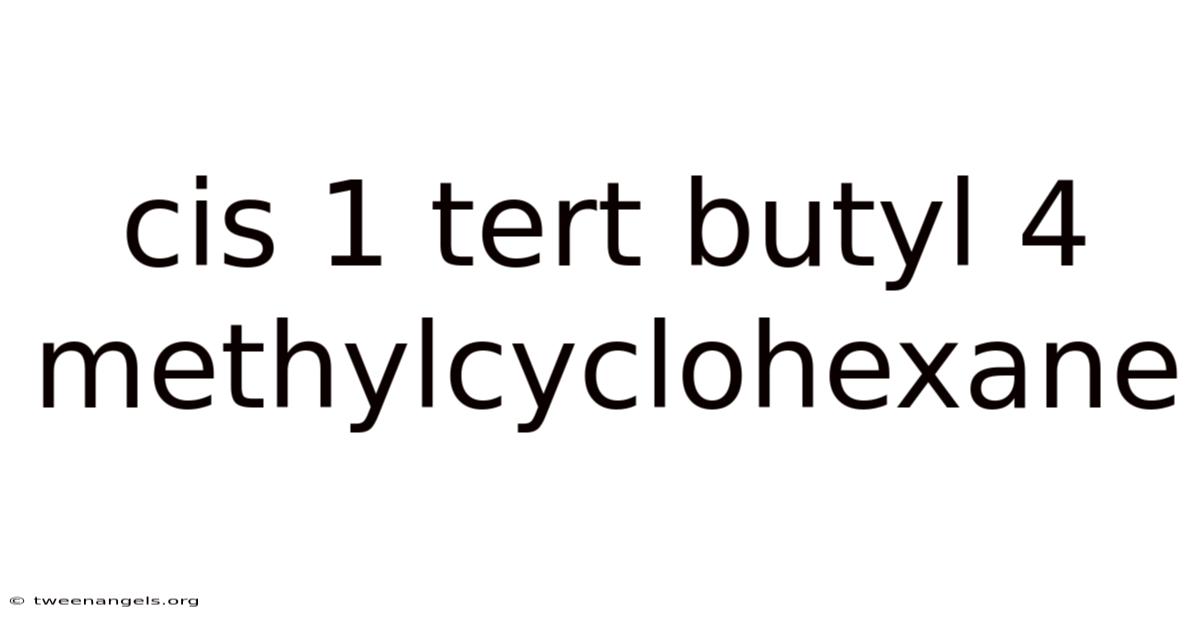
Table of Contents
cis-1-tert-butyl-4-methylcyclohexane represents a fascinating example of how molecular structure dictates stability and reactivity within the realm of organic chemistry. This specific stereoisomer, characterized by its cis configuration at the 1 and 4 positions on the cyclohexane ring, offers a compelling case study in stereochemistry, conformational analysis, and the influence of bulky substituents. Understanding its properties provides crucial insights into the behavior of substituted cyclohexanes and the principles governing molecular geometry.
Introduction Cyclohexane rings, with their alternating single and double bond character in the chair conformation, form the backbone of numerous natural and synthetic compounds. When substituents are attached to these rings, their spatial arrangement relative to each other becomes paramount. cis-1-tert-butyl-4-methylcyclohexane is a prime example. The "cis" designation indicates that the tert-butyl group (a bulky, branched alkyl group) and the methyl group (a small alkyl group) are oriented on the same face of the ring. This seemingly simple configuration has profound implications for the molecule's energy profile, stability, and potential reactivity pathways. This article delves into the synthesis, structure, conformational behavior, and significance of cis-1-tert-butyl-4-methylcyclohexane, highlighting why its specific arrangement is both chemically relevant and energetically distinct.
Synthesis: Building the Molecule The synthesis of cis-1-tert-butyl-4-methylcyclohexane typically involves the functionalization of a cyclohexyl precursor. A common approach begins with cyclohexyl chloride (or another suitable halide). This compound undergoes nucleophilic substitution. To achieve the cis stereochemistry at the 1 and 4 positions, the reaction conditions and the nature of the nucleophile are crucial. Direct substitution at the 1-position on the cyclohexyl ring is challenging due to ring strain. Often, a more feasible route involves:
- Functionalization: First, a substituent is introduced at the 4-position. For example, cyclohexyl chloride can be converted to cyclohexyl methyl ether (via Williamson ether synthesis with methanol and K₂CO₃), creating a 4-methylcyclohexyl group.
- Substitution at Position 1: The 4-methylcyclohexyl group is then reacted with tert-butyl chloride in the presence of a base like triethylamine. This substitution occurs at the less hindered 1-position of the cyclohexane ring. Crucially, if the reaction is carried out under conditions favoring retention of configuration (e.g., using a mild base and controlling solvent polarity), the stereochemistry at the 1-position relative to the existing 4-methyl group can be controlled to be cis. This might involve careful choice of reaction conditions or the use of specific catalysts to favor the desired stereoisomer formation. The resulting product is cis-1-tert-butyl-4-methylcyclohexane.
Scientific Explanation: Structure, Conformation, and Stability The cis configuration in cis-1-tert-butyl-4-methylcyclohexane is inherently linked to the molecule's conformational preferences and energy landscape. The cyclohexane ring adopts its most stable chair conformation, where all carbon-hydrogen bonds are staggered.
- Conformational Analysis: In the chair conformation, substituents at positions 1 and 4 can be either axial or equatorial. The cis configuration means that if the tert-butyl group is equatorial at position 1, the methyl group must be axial at position 4 (and vice-versa), and vice-versa. This is because adjacent positions in a cis-disubstituted cyclohexane alternate between axial and equatorial orientations in the chair flip.
- Steric Strain: The key factor distinguishing cis-1-tert-butyl-4-methylcyclohexane from its trans counterpart is the steric interaction between the bulky tert-butyl group and the axial substituent at position 4 (or vice-versa). The tert-butyl group is exceptionally large (a tertiary butyl group is approximately 1.7 Å in radius). When it occupies an axial position (as it must when the methyl is equatorial, or vice-versa), it experiences significant steric repulsion with the axial hydrogens or substituents on the adjacent carbons (C3 and C5). This results in a higher energy conformation for the cis isomer compared to the trans isomer.
- Trans Isomer Advantage: The trans isomer (cis-1-tert-butyl-4-methylcyclohexane is a misnomer; it should be called trans-1-tert-butyl-4-methylcyclohexane) has the tert-butyl and methyl groups on opposite faces. In the chair conformation, this allows both groups to be equatorial simultaneously. This minimizes steric strain, as the large tert-butyl group avoids the axial interactions. Consequently, the trans isomer is significantly more stable than the cis isomer. The energy difference between the two isomers is a classic example of how steric bulk influences conformational preference and thermodynamic stability in substituted cyclohexanes.
- Ring Flip Dynamics: The cis isomer can undergo ring flip, interconverting between its two chair conformations (one with tert-butyl equatorial and methyl axial, and the other with tert-butyl axial and methyl equatorial). However, both chair conformations of the cis isomer are higher in energy than either chair conformation of the trans isomer due to the unavoidable axial tert-butyl interaction in one chair form. The trans isomer also undergoes ring flip, interconverting between two chair conformations,
The trans isomer also undergoes ring flip, interconverting between two chair conformations, each of which places the bulky tert‑butyl group in an equatorial position while the methyl remains equatorial on the opposite face of the ring. Because both chairs are low‑energy, the trans system exhibits a single, rapidly equilibrating conformer at ambient temperature; interconversion is essentially invisible by most spectroscopic techniques. In contrast, the cis isomer populates two distinct chair forms that are both higher in energy, and the equilibrium between them is temperature‑dependent. At low temperature the axial‑tert‑butyl conformer dominates, giving rise to characteristic chemical‑shift differences for the axial methyl protons in the ¹H‑NMR spectrum, whereas at elevated temperatures the axial/equatorial exchange accelerates, broadening the signals and reflecting the dynamic nature of the conformational landscape.
Quantitative studies employing variable‑temperature ¹H‑NMR and computational methods (such as MP2 and DFT with appropriate dispersion corrections) have yielded an experimentally measured free‑energy difference (ΔG°) of approximately 2.5–3.0 kcal mol⁻¹ favoring the trans isomer under standard conditions. This magnitude aligns closely with the calculated steric penalty associated with forcing a tert‑butyl group into an axial position, underscoring the dominance of steric effects over electronic or hyperconjugative contributions in this system. Moreover, the trans isomer’s superior stability translates into a higher melting point and lower solubility in non‑polar solvents, properties that are routinely exploited in synthetic planning when a chemist seeks a more robust, less sterically congested scaffold.
Beyond pure thermodynamics, the conformational behavior of cis‑1‑tert‑butyl‑4‑methylcyclohexane offers a vivid illustration of how steric bulk can dictate reactivity. In substitution reactions that proceed through a chair‑like transition state, the axial orientation of the tert‑butyl group can hinder nucleophilic attack at the adjacent carbon, leading to altered regio‑ and stereochemical outcomes compared with the trans analogue. Likewise, oxidation or functional‑group interconversion steps that require a specific orientation of the substituents can be steered by the conformational equilibrium, allowing researchers to exploit the slower interconversion of the cis system as a means of kinetic control.
In summary, the conformational preferences of cis‑1‑tert‑butyl‑4‑methylcyclohexane are governed by the inevitable clash that arises when a large tert‑butyl group occupies an axial position. This clash raises the energy of every chair conformer relative to the trans counterpart, where both substituents can reside equatorially. The resulting energy gap manifests in measurable differences in spectroscopic signatures, thermodynamic stability, and reactivity patterns. Recognizing these subtle yet profound steric effects not only deepens our understanding of cyclohexane conformational analysis but also equips synthetic chemists with a predictive tool for designing molecules in which steric demand dictates overall molecular architecture and behavior.
The conformational preferences of cis-1-tert-butyl-4-methylcyclohexane are governed by the inevitable clash that arises when a large tert-butyl group occupies an axial position. This clash raises the energy of every chair conformer relative to the trans counterpart, where both substituents can reside equatorially. The resulting energy gap manifests in measurable differences in spectroscopic signatures, thermodynamic stability, and reactivity patterns. Recognizing these subtle yet profound steric effects not only deepens our understanding of cyclohexane conformational analysis but also equips synthetic chemists with a predictive tool for designing molecules in which steric demand dictates overall molecular architecture and behavior. By appreciating how the cis arrangement forces one bulky group into an energetically unfavorable axial orientation, researchers can anticipate and exploit these conformational biases to control selectivity, reactivity, and physical properties in complex synthetic targets.
Latest Posts
Latest Posts
-
Fundamentals Of Anatomy And Physiology 12th Edition
Mar 15, 2026
-
Which Organelle Is Not Found In An Animal Cell
Mar 15, 2026
-
Assessing Learners With Special Needs An Applied Approach
Mar 15, 2026
-
The African American Odyssey Combined Volume
Mar 15, 2026
-
A Topical Approach To Lifespan Development
Mar 15, 2026
Related Post
Thank you for visiting our website which covers about Cis 1 Tert Butyl 4 Methylcyclohexane . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.