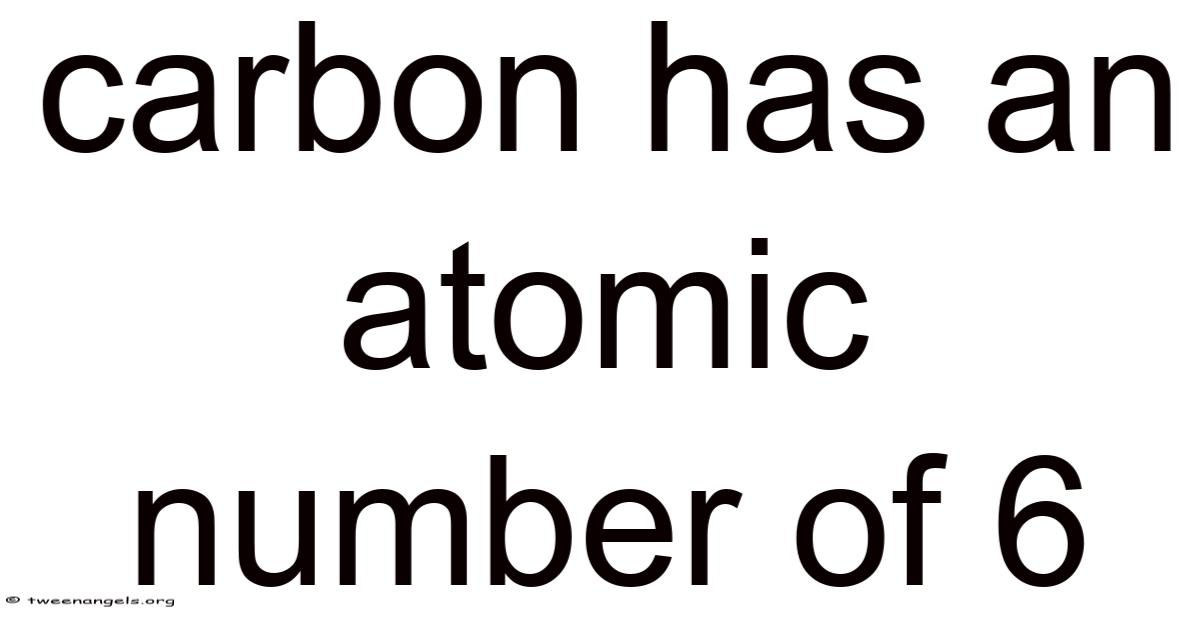Carbon has an atomic number of 6, and this single fact unlocks an extraordinary world of chemistry, biology, and material science. Understanding why carbon carries this specific atomic number — and what that number means — is the key to appreciating one of the most important elements in the universe. From the diamonds on your finger to the DNA in your cells, carbon is everywhere, and it all starts with those six protons at its core.
What Is an Atomic Number?
Before diving into the specifics of carbon, Understand what an atomic number actually represents — this one isn't optional. Which means the atomic number of an element is defined as the number of protons found in the nucleus of a single atom of that element. This number is unique to each element and serves as its defining characteristic on the periodic table of elements Most people skip this — try not to. That's the whole idea..
Here are the key points about atomic numbers:
- The atomic number determines the chemical identity of an element.
- It tells you exactly how many protons exist in the nucleus.
- In a neutral atom, the atomic number also equals the number of electrons orbiting the nucleus.
- Elements are arranged on the periodic table in ascending order of their atomic numbers.
Take this: hydrogen has an atomic number of 1 (one proton), helium has an atomic number of 2 (two protons), and so on. When we say that carbon has an atomic number of 6, we are stating an unchangeable truth about its nuclear composition: every carbon atom contains exactly six protons It's one of those things that adds up..
Carbon's Atomic Number and What It Means
The fact that carbon has an atomic number of 6 carries profound implications. Let us break this down:
-
Six protons in the nucleus — This is the defining feature. Remove even one proton, and the atom would no longer be carbon; it would become boron (atomic number 5). Add one proton, and it becomes nitrogen (atomic number 7).
-
Six electrons in a neutral atom — Because atoms are electrically neutral, the number of negatively charged electrons must balance the positive charge of the protons. So, a neutral carbon atom has six electrons distributed across energy levels.
-
Six as a "magic" number for bonding — Carbon's six electrons are arranged in two energy levels: two electrons fill the first shell, and four electrons occupy the second shell. This configuration is critically important because carbon needs four more electrons to complete its outer shell, making it exceptionally versatile in forming chemical bonds.
The Structure of a Carbon Atom
To fully grasp why carbon has an atomic number of 6 and why that matters, we need to examine the atom's internal structure.
Protons and Neutrons
The nucleus of a carbon atom contains six protons and typically six neutrons. Together, they account for nearly all of the atom's mass. The protons carry a positive charge, while neutrons are electrically neutral. Think about it: the total number of protons and neutrons is called the mass number. For the most common isotope of carbon (carbon-12), the mass number is 12 (6 protons + 6 neutrons).
And yeah — that's actually more nuanced than it sounds.
Electron Configuration
Carbon's six electrons are arranged in the following configuration:
- First energy level (K shell): 2 electrons
- Second energy level (L shell): 4 electrons
The second energy level can hold up to 8 electrons. Because carbon has only 4 electrons in this outer shell, it is not stable on its own. This incomplete outer shell drives carbon's remarkable tendency to form bonds with other atoms, including other carbon atoms.
Covalent Bonding Ability
Because carbon has four valence electrons, it can form up to four covalent bonds. This property is virtually unmatched among the lighter elements and is the reason carbon can create an immense variety of molecular structures — from simple methane (CH₄) to incredibly complex biological macromolecules like proteins and nucleic acids.
Why Carbon's Atomic Number Matters
The significance of carbon having an atomic number of 6 extends far beyond a textbook fact. Here is why it matters:
1. Foundation of Organic Chemistry
Organic chemistry is the branch of chemistry dedicated to the study of carbon-containing compounds. That's why carbon's ability to form stable covalent bonds with itself and other elements gives rise to millions of known organic compounds. Without those six protons — and the resulting six-electron configuration — none of this would be possible.
Easier said than done, but still worth knowing.
2. Basis of Life
Every known form of life on Earth is built on carbon-based molecules. DNA, RNA, carbohydrates, lipids, and proteins all rely on carbon's bonding versatility. The atomic number of 6 essentially makes carbon the backbone of biological complexity.
3. Material Diversity
Carbon's atomic structure allows it to form radically different materials depending on how its atoms bond:
- Diamond — Each carbon atom is bonded to four others in a rigid tetrahedral lattice, creating the hardest known natural material.
- Graphite — Carbon atoms form layers of hexagonal rings that slide over each other, making graphite soft and ideal for use in pencils and lubricants.
- Graphene — A single layer of graphite, graphene is one of the strongest and most conductive materials known to science.
- Fullerenes and Carbon Nanotubes — These exotic structures have unique electrical and mechanical properties being explored in nanotechnology.
All of this diversity traces back to the same six protons in the nucleus It's one of those things that adds up..
Isotopes of Carbon
While every carbon atom has 6 protons (because that is what defines it as carbon), the number of neutrons can vary. These variations are called isotopes.
| Isotope | Protons | Neutrons | Mass Number | Stability |
|---|---|---|---|---|
| Carbon-12 | 6 | 6 | 12 | Stable |
| Carbon-13 | 6 | 7 | 13 | Stable |
| Carbon-14 | 6 | 8 | 14 | Radioactive |
- Carbon-12 is by far the most abundant isotope, making up about 98.9% of all naturally occurring carbon. It serves as the standard against which atomic masses are measured.
- Carbon-13 is also stable and is used in scientific research, particularly in nuclear magnetic resonance (NMR) spectroscopy.
- Carbon-14 is radioactive and decays over time. This property makes it invaluable in radiocarbon dating, a technique used to determine the age of archaeological and geological samples up to about 50,000 years old.
The existence of these isotopes demonstrates that while the atomic number (6 protons) defines the element, the neutron count can vary without changing the element's identity.
Carbon in the Periodic Table
On the periodic table, carbon sits in Group 14 (also known as the carbon group) and Period 2. Its position tells us a lot:
- Group 14 means carbon has four valence electrons, shared by silicon (Si), germanium (Ge), tin (Sn), and lead (Pb).
- Period 2 indicates that carbon's electrons occupy the second energy level, making it a
Carbon’s unique role in sustaining life and advancing technology stems from its ability to form an astonishing array of compounds, all anchored by its carbon backbone. Think about it: this versatility is mirrored in the periodic table, where carbon’s placement and properties set the stage for its participation in diverse chemical reactions. From the delicate rings of graphite to the crystalline sheets of graphite, and from the detailed structures of DNA to the strong frameworks of diamond, each form highlights carbon’s adaptability. The periodic table not only classifies these forms but also reveals how subtle changes in electron arrangement can lead to radically different materials.
Beyond that, understanding carbon’s isotopes deepens our appreciation of its behavior over time. Plus, while stable carbon-12 dominates natural samples, the presence of carbon-14 offers a window into Earth’s ancient past, helping scientists unravel history hidden within the rings of trees and the layers of rock. This interplay between stability and transformation underscores carbon’s enduring significance in both science and society.
In essence, carbon’s atomic characteristics, isotopic variations, and material diversity form a cohesive narrative that shapes our world—from the smallest molecules to the grandest technological innovations. Worth adding: the periodic table serves as a guiding compass, connecting these concepts into a unified understanding of nature’s design. Recognizing this interconnectedness not only enriches our knowledge but also inspires further exploration into the mysteries of carbon’s role in life and technology.
Conclusion: Carbon’s unparalleled versatility, governed by its atomic structure and isotopic diversity, makes it indispensable to both biological systems and human advancements. Its presence in the periodic table reinforces its foundational role, bridging the microscopic with the macroscopic in ways that continue to captivate scientists and innovators alike.