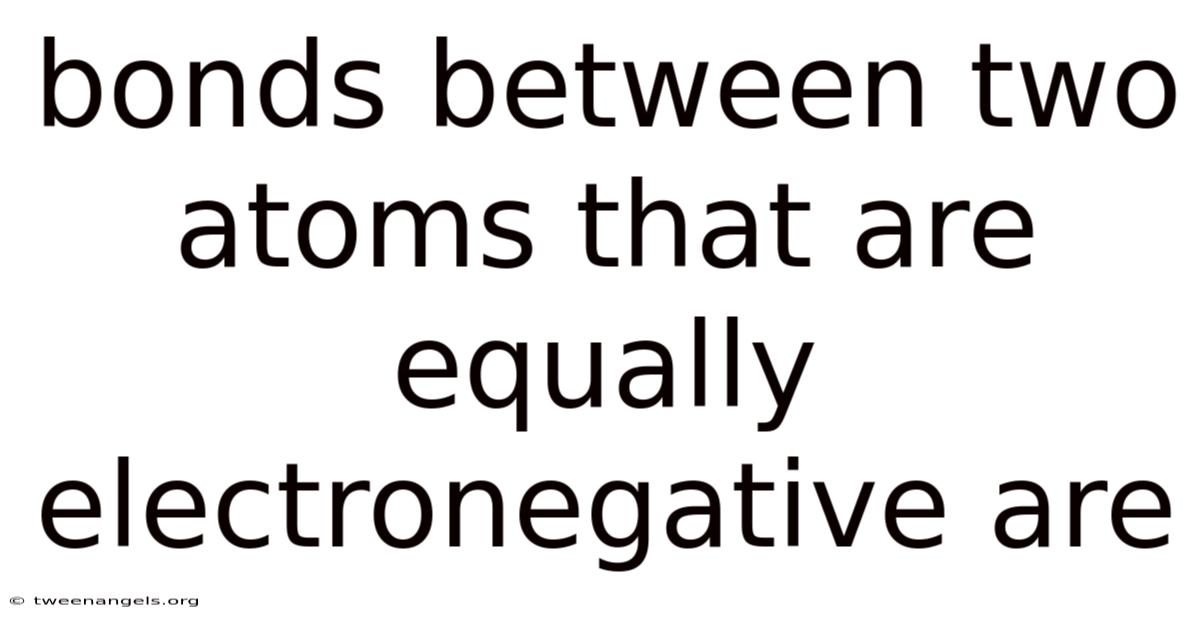The detailed dance of atomic interactions underpins the very fabric of matter, revealing a universe where precision governs creation and consequence. At the heart of this phenomenon lies the concept of bonds formed between two atoms possessing equal electronegativity—a principle that defies intuition yet governs the stability of molecular structures. These bonds emerge not through dramatic shifts in charge distribution but through a delicate balance of shared and retained electrons, offering a window into the hidden forces shaping the cosmos. Understanding this equilibrium requires delving into the nuances of atomic behavior, where even minor deviations can cascade into profound implications for chemistry, physics, and biology. Such knowledge serves as a cornerstone for scientists seeking to unravel the mysteries of molecular biology, materials science, and environmental chemistry, all of which hinge on the subtle interplay of electron affinity and atomic properties. In this exploration, we will dissect the mechanics behind such bonds, examining their role in shaping everything from the molecular weight of compounds to the very properties of substances like water and carbon dioxide. The journey begins with defining electronegativity itself, a concept that demands careful scrutiny to avoid misconceptions. Practically speaking, while often associated with polarity, electronegativity is more accurately a measure of an atom’s capacity to attract electrons within a bond, a distinction that distinguishes it from other properties like ionization energy or atomic radius. This leads to this foundational understanding sets the stage for appreciating how two atoms of comparable strength can forge a bond that is both stable and predictable, yet remains a subject of ongoing research due to its complexities. The implications of such bonds extend beyond the laboratory, influencing everything from industrial processes to natural ecosystems, making their study a vital pursuit for both academic and practical applications. Think about it: as we delve deeper, it becomes evident that the equilibrium between equal electronegativity is not merely a theoretical construct but a practical reality that defines the behavior of countless substances. This article will traverse the landscape of covalent bonding, explore its manifestations through various examples, and examine its significance in advancing scientific knowledge. Through this process, we aim to illuminate the profound connection between atomic properties and the macroscopic world around us, offering insights that transcend mere academic interest to become essential tools for addressing real-world challenges.
Real talk — this step gets skipped all the time.
Covalent bonds formed between atoms of equal electronegativity represent a paradigm shift in how we conceptualize chemical interactions. Still, the study of equal electronegativity bonds also raises intriguing questions about their role in environmental systems. Think about it: such bonds are particularly significant in biochemistry, where proteins and enzymes depend on precise molecular interactions to function effectively. On top of that, additionally, the prevalence of these bonds in natural substances such as diamond (C₄C₄) highlights their historical and geological significance, as diamond’s stability stems from carbon atoms sharing electrons equally, making it a testament to the durability of covalent networks. Practically speaking, this symmetry not only enhances stability but also results in predictable molecular geometries, such as tetrahedral arrangements in methane (CH₄) or linear structures in diatomic molecules like O₂. These nuances necessitate a nuanced approach when applying theoretical models to practical applications, prompting scientists to refine their understanding continuously. Worth adding, the stability of these bonds influences reaction rates, as molecules with stronger covalent ties often exhibit higher thermal resistance and lower reactivity compared to those with weaker or polar bonds. Still, unlike polar covalent bonds, where electron sharing is uneven due to disparities in electronegativity, equal electronegativity ensures a symmetrical distribution, leading to bonds characterized by minimal repulsion between electron pairs. While the idealized model of equal electronegativity assumes perfect symmetry, real-world scenarios often introduce variables such as hybridization, molecular vibrations, or solvent effects that slightly perturb the equilibrium. This property is critical in synthesizing pharmaceuticals, where controlled bond strength ensures efficacy and safety. These bonds arise when two atoms share electrons nearly equally, resulting in a stable configuration where each atom retains a fraction of the shared electrons, thereby minimizing energy expenditure. Take this: the interaction between such molecules in atmospheric chemistry can affect climate patterns by altering the behavior of gases like nitrogen and oxygen, which collectively contribute to Earth’s greenhouse effect. What's more, the study of such bonds extends beyond chemistry into fields like materials engineering, where designing materials with specific properties—such as high thermal conductivity or chemical resistance—requires meticulous consideration of atomic interactions. Still, yet, even within these seemingly straightforward bonds, subtleties emerge. Here's a good example: the formation of peptide bonds in amino acids relies on identical electronegativity between carbon, nitrogen, and oxygen atoms, ensuring the integrity of the polypeptide chain. The absence of significant charge separation in these bonds underscores their role as the foundation for many essential chemical compounds, including water (H₂O), which relies on oxygen’s ability to share electrons with hydrogen without inducing substantial polarization. In this context, the importance of equal electronegativity bonds transcends academic curiosity, becoming a practical imperative for innovation across disciplines.
...powerful lens through which to understand the fundamental building blocks of matter and the complex processes that govern our world.
At the end of the day, the concept of equal electronegativity in covalent bonds, while seemingly simple, unveils a profound level of complexity and importance. On the flip side, it underpins the stability of life-sustaining molecules, dictates the efficacy of pharmaceutical interventions, and influences global environmental dynamics. From the diamond’s enduring strength to the delicate dance of atmospheric gases, these bonds are integral to the universe as we know it. While theoretical models offer valuable insights, the ever-present influence of real-world variables demands continuous refinement and a multidisciplinary approach to fully grasp their intricacies. That said, ultimately, the continued exploration of equal electronegativity bonds promises to reach further advancements in materials science, chemical engineering, and our fundamental understanding of the chemical world, driving innovation and addressing critical challenges facing society. The pursuit of understanding these seemingly straightforward interactions is not merely an academic exercise; it's a vital endeavor with far-reaching implications for the future Simple, but easy to overlook. And it works..
The Earth’s greenhouse effect remains a cornerstone of climate science, shaping our planet’s temperature and weather patterns through complex interactions involving gases like carbon dioxide and methane. As we delve deeper into this phenomenon, it becomes clear how interconnected it is with the molecular foundations of our environment. Because of that, the prevalence of stable bonds in natural substances, such as diamond, further underscores the significance of these interactions. Diamond’s structure, built from carbon atoms sharing electrons equally, exemplifies the power of covalent networks in creating enduring materials. Yet, this stability is not without its challenges; real-world conditions—such as temperature fluctuations or structural stress—can subtly shift equilibrium, reminding us that even in seemingly perfect bonds, external factors play a crucial role. This interplay between ideal theory and practical reality emphasizes the need for ongoing research and adaptation in scientific understanding. In the broader scope, these bonds extend beyond laboratories, influencing everything from industrial materials to ecological systems. Their study not only reveals the resilience of certain structures but also highlights the delicate balance required to sustain life and environmental stability. As we continue to explore these relationships, we move closer to harnessing the potential of chemical interactions for sustainable progress. At the end of the day, this journey reinforces the idea that even the simplest bonds carry profound implications, shaping both our knowledge and our future.
The study of these molecular interactions is not only a testament to scientific curiosity but also a catalyst for innovation across disciplines. Consider this: the pursuit of this knowledge bridges the gap between abstract theory and tangible applications, reinforcing the importance of a holistic approach. In real terms, by examining the strength and behavior of bonds at various scales, researchers can better design materials that withstand harsh conditions, improve energy storage solutions, and develop cleaner technologies. As we continue to refine our understanding, each discovery builds upon the last, weaving a richer tapestry of possibilities. This dynamic process underscores how foundational principles guide progress, shaping not just laboratory findings but also real-world outcomes It's one of those things that adds up..
The influence of such bonds extends beyond the physical realm, affecting ecological systems and human health in subtle yet significant ways. On top of that, grasping their complexities enables us to anticipate environmental shifts and mitigate risks, fostering resilience in an ever-changing world. This ongoing exploration highlights the interconnectedness of science, technology, and sustainability, reminding us that every bond, no matter how small, plays a role in the greater scheme Practical, not theoretical..
Simply put, delving deeper into the nature of these interactions empowers us to address pressing challenges and envision a future guided by informed insight. The journey continues, propelled by the relentless quest to decode and harness the language of chemistry. This commitment not only advances our intellectual frontiers but also strengthens our capacity to contribute meaningfully to society’s needs Worth keeping that in mind..
Conclusion: The exploration of molecular bonds remains a cornerstone of scientific advancement, underscoring the profound impact of fundamental research on our collective future. Each step forward highlights the power of curiosity and collaboration in shaping a sustainable and innovative world That's the whole idea..