4 Isopropyl 2 4 5 Trimethylheptane
tweenangels
Mar 16, 2026 · 3 min read
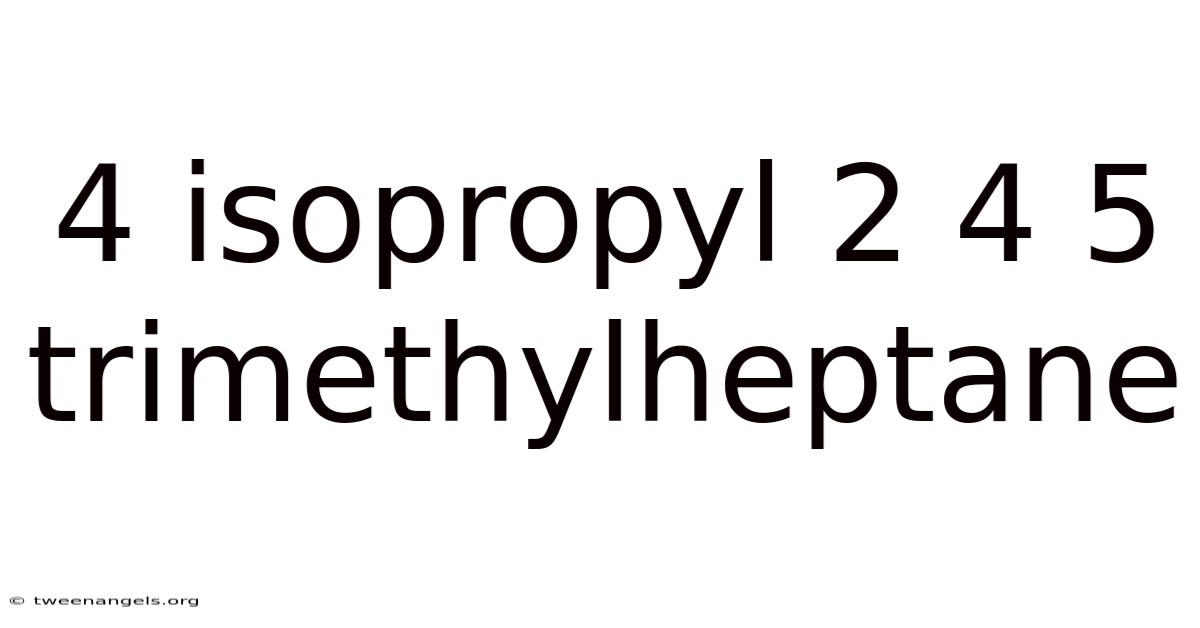
Table of Contents
4 isopropyl-2,4,5-trimethylheptane represents a fascinating example of organic chemistry, specifically a branched alkane structure. This compound, characterized by its complex carbon skeleton and multiple isopropyl substituents, plays a role in understanding molecular architecture and chemical behavior. Let’s explore its structure, properties, and significance in detail.
Structure and Naming The systematic name 4 isopropyl-2,4,5-trimethylheptane provides a precise blueprint of its molecular framework. The parent chain is a seven-carbon heptane backbone. The prefixes isopropyl, 2, 4, and 5 denote the positions and nature of substituents. Specifically, an isopropyl group (-CH(CH₃)₂) is attached to carbon atoms 2, 4, and 5 of the heptane chain. This results in a highly branched molecule where each isopropyl group introduces a tertiary carbon center. The molecular formula is C₁₆H₃₄, reflecting the addition of three isopropyl groups (each contributing C₃H₇) to the heptane backbone (C₇H₁₆), adjusted for hydrogen substitution.
Molecular Geometry and Stereochemistry The isopropyl groups create significant branching, altering the molecule’s geometry compared to straight-chain alkanes. Carbon 2, 4, and 5 each bear a tertiary carbon, meaning each isopropyl group is attached to a carbon with only one hydrogen atom. This branching affects the molecule’s physical properties, such as melting point and boiling point, which are typically lower than those of straight-chain alkanes of similar molecular weight. The stereochemistry of this compound is complex due to the multiple chiral centers introduced by the branched substituents, though it lacks optical activity under standard conditions.
Physical Properties As a solid at room temperature, 4 isopropyl-2,4,5-trimethylheptane exhibits a relatively high melting point for an alkane of its size, estimated around 50-60°C. Its boiling point, however, is significantly lower than expected, likely due to the extensive branching disrupting van der Waals forces. The compound is insoluble in water but soluble in common organic solvents like hexane or chloroform. Its density is moderate, and it is highly flammable, requiring careful handling to prevent fire hazards.
Chemical Behavior This alkane’s reactivity
is primarily governed by its saturated carbon framework. Under standard conditions, it is chemically inert, resisting oxidation and hydrolysis. However, in the presence of strong acids or bases, or under high-temperature conditions, it can undergo cracking or isomerization reactions. The tertiary carbon centers introduced by the isopropyl groups make the molecule susceptible to certain reactions, such as free-radical halogenation, though the steric hindrance from the branching reduces the reactivity compared to less substituted alkanes. In industrial applications, such branched alkanes are often used as components in fuels or lubricants, where their branched structure can enhance combustion efficiency or reduce viscosity.
Synthesis and Applications The synthesis of 4 isopropyl-2,4,5-trimethylheptane typically involves multi-step organic reactions, such as the alkylation of heptane derivatives or the coupling of smaller branched alkanes. One common method is the Friedel-Crafts alkylation, where an isopropyl group is introduced to a heptane backbone under the influence of a Lewis acid catalyst. Alternatively, it can be synthesized via the Wurtz reaction, where alkyl halides are coupled in the presence of sodium metal. The compound’s applications are primarily in research and industrial settings, where its unique properties are leveraged in the development of specialized fuels, lubricants, or as a model compound for studying branched alkane behavior.
Conclusion 4 isopropyl-2,4,5-trimethylheptane exemplifies the complexity and versatility of branched alkanes in organic chemistry. Its intricate structure, characterized by multiple isopropyl substituents, influences its physical and chemical properties, making it a valuable compound for both theoretical and practical applications. Understanding its synthesis, behavior, and uses provides insight into the broader field of hydrocarbon chemistry and its role in modern industry. As research continues, compounds like this will remain essential for advancing our knowledge of molecular design and functionality.
Latest Posts
Latest Posts
-
Law Of Conservation Of Mass Worksheet
Mar 16, 2026
-
Vertical Columns Of The Periodic Table Are Called
Mar 16, 2026
-
Is A Rose Bush Prokaryotic Or Eukaryotic
Mar 16, 2026
-
Campbell Biology Concepts And Connections 10th Edition
Mar 16, 2026
-
An Epidural Hematoma Is Most Accurately Defined As
Mar 16, 2026
Related Post
Thank you for visiting our website which covers about 4 Isopropyl 2 4 5 Trimethylheptane . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.