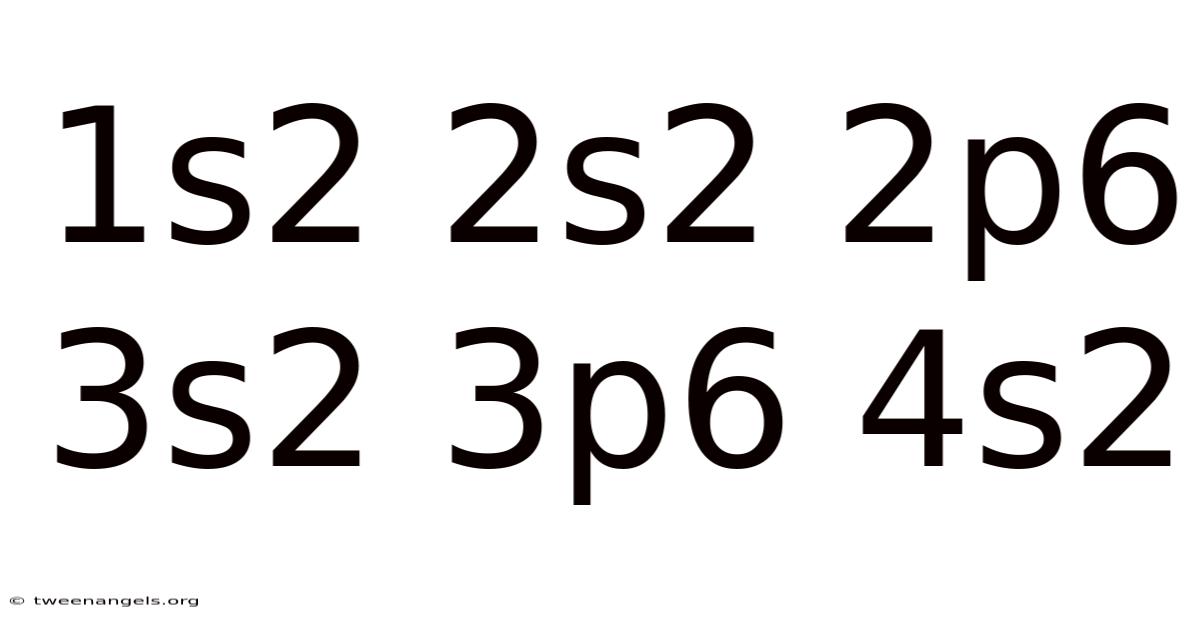The intricate dance of particles within atomic nuclei and the delicate orchestration of electrons within their respective shells has long fascinated scientists and curious minds alike. At the heart of this cosmic ballet lies the concept of electron configurations, a foundational pillar in understanding the behavior of matter at the atomic level. These configurations dictate everything from an element’s physical properties to its chemical reactivity, shaping the very fabric of the universe around us. Whether explaining why certain elements form specific compounds or why certain atoms emit light in distinct wavelengths, electron configurations serve as the cornerstone of chemical theory and practical applications. This article delves deeply into the principles behind these arrangements, exploring their significance across disciplines while emphasizing their practical relevance. Through this exploration, readers will uncover how the arrangement of electrons not only defines an atom’s identity but also influences its interactions with other substances, making electron configurations a vital tool for scientists, educators, and even everyday problem-solvers alike. The complexity inherent in these arrangements often masks a profound simplicity, requiring careful attention to detail yet offering immense utility when mastered. As we unravel this intricate subject, it becomes evident that mastering electron configurations is akin to deciphering the language of the cosmos itself, revealing patterns and relationships that underpin much of nature’s structure.
Electron configurations act as a universal blueprint for understanding atomic behavior, serving as the basis for predicting how elements interact. At their core, these configurations describe how electrons distribute themselves among the different energy levels or shells surrounding a nucleus. Each shell corresponds to a specific energy level, with subshells further subdivided into orbitals that house pairs of electrons. The filling order—descending by energy or following the Aufbau principle—provides a systematic framework for constructing these arrangements. For instance, the familiar noble gases like neon or argon exhibit full valence shells, reflecting their stability and tendency to gain or lose electrons. Conversely, transition metals exhibit complex configurations due to partially filled d-orbitals, influencing their reactivity and properties. Such patterns are not arbitrary; they arise from quantum mechanical principles that govern electron behavior, balancing stability with the pursuit of lower energy states. This balance is central to the periodic table’s organization, where elements are grouped by similar configurations, enabling patterns in chemical properties that define their behavior in reactions. The study of these configurations thus bridges theoretical knowledge with observable phenomena, offering a lens through which to interpret the tangible world. Furthermore, deviations from standard configurations, such as those seen in some organic molecules or excited states, highlight the dynamic nature of atomic systems and their responsiveness to external influences. Such flexibility underscores the importance of electron configurations in addressing challenges ranging from material science to biochemistry, where precise control over atomic arrangements can lead to breakthroughs.
One of the most compelling aspects of electron configurations is their role in predicting chemical reactivity. Elements with similar configurations often exhibit comparable reactivity, while those differing by a single electron or orbital can exhibit distinct behaviors. For example, alkali metals like sodium and potassium share electron configurations that place their valence electrons in the outermost shell, leading to high reactivity with water and other substances. In contrast, halogens such as fluorine and chlorine possess configurations that allow them to readily gain electrons, forming strong bonds with metals. This principle extends beyond the periodic table, influencing how compounds form and interact within molecular structures. Additionally, the presence of certain subshells, like the f-orbitals in lanthanides or actinides, introduces complexity that can significantly alter an element’s chemical properties, necessitating nuanced understanding. Such nuances are critical in fields like pharmaceuticals, where precise molecular design relies on controlled electron distributions. Even in inorganic chemistry, the configuration of
...elements dictates the stability and properties of compounds, impacting everything from the hardness of ceramics to the conductivity of semiconductors.
Beyond reactivity, electron configurations underpin a vast range of physical properties. The energy levels of electrons within an atom determine its ionization energy – the energy required to remove an electron. This, in turn, influences an element's ability to form ions and participate in chemical bonding. Similarly, the electron configuration dictates an element's atomic radius, with electrons occupying lower energy levels resulting in smaller, more compact atoms. These properties are not merely theoretical curiosities; they are fundamental to understanding the behavior of matter and designing materials with specific characteristics.
The ongoing exploration of electron configurations continues to yield exciting discoveries. Advanced computational methods are enabling researchers to predict and understand complex molecular structures with unprecedented accuracy. This allows for the rational design of new materials with tailored properties, such as high-temperature superconductors or efficient solar cells. Furthermore, advancements in spectroscopic techniques provide direct insights into electron behavior, furthering our understanding of chemical bonding and molecular interactions.
In conclusion, the electron configuration is not just a fundamental concept in chemistry; it is the bedrock upon which the periodic table is built and the key to unlocking the secrets of matter. From predicting chemical reactivity and physical properties to driving innovation in materials science and medicine, the study of electron configurations offers a profound and continuously evolving perspective on the world around us. As our understanding deepens, we can anticipate even more transformative applications emerging from this corner of scientific inquiry, ultimately leading to a more technologically advanced and sustainable future.
Continuing from the establishedfoundation, the profound influence of electron configurations extends far beyond the traditional realms of chemistry and materials science, permeating into the very fabric of modern technology and our understanding of the universe. This intricate dance of electrons dictates not only the behavior of atoms and molecules but also the fundamental interactions that govern complex systems, from the quantum computing architectures of tomorrow to the biochemical pathways sustaining life.
Consider the burgeoning field of quantum information science. Here, the spin states of electrons – a direct consequence of their specific configurations – form the basis of qubits, the fundamental units of quantum computers. The precise control and manipulation of these spin states, governed by the underlying electron configuration, are paramount for achieving quantum coherence and enabling computations beyond the reach of classical machines. Similarly, in nanotechnology, the electronic structure determines the optical and electrical properties of nanomaterials, allowing for the design of sensors, catalysts, and transistors with unprecedented efficiency and functionality.
Furthermore, electron configurations play a critical role in understanding and harnessing energy at its most fundamental level. The intricate electron arrangements within materials dictate their ability to conduct electricity, absorb light, or store energy. This knowledge is essential for developing next-generation solar cells with higher efficiencies, batteries with greater capacity and longer lifespans, and superconductors operating at higher temperatures, all of which are vital for a sustainable energy future. The quest to manipulate electron configurations for optimal energy conversion and storage drives significant research in condensed matter physics and materials chemistry.
The exploration of electron configurations also deepens our comprehension of cosmic phenomena. In astrophysics, the electron configurations of elements in stellar atmospheres and interstellar clouds determine their spectral signatures, allowing astronomers to probe the composition, temperature, and dynamics of distant celestial bodies. Understanding how electron configurations influence atomic and molecular behavior under extreme conditions of pressure and temperature found within planets and stars provides crucial insights into the formation and evolution of planetary systems and galaxies.
In essence, the electron configuration remains the quintessential blueprint for matter. Its mastery unlocks the potential to engineer matter at the atomic scale, revolutionizing fields as diverse as medicine, computing, energy, and space exploration. As computational power and experimental techniques continue to advance, our ability to predict, control, and exploit these configurations grows exponentially, promising transformative breakthroughs that will reshape our technological landscape and deepen our understanding of the cosmos. The journey to decode the full implications of electron configurations is far from complete, yet it stands as one of the most exhilarating frontiers in scientific inquiry, holding the keys to a future defined by unprecedented innovation and discovery.
Conclusion:
The electron configuration is far more than a theoretical construct; it is the fundamental language of matter, dictating the very essence of chemical and physical behavior across the cosmos. From the intricate design of pharmaceuticals and the robustness of ceramics to the efficiency of solar cells and the power of quantum computers, our ability to understand and manipulate this atomic blueprint is the cornerstone of scientific progress and technological advancement. As we continue to unravel its complexities through ever-more sophisticated computational models and experimental probes, the potential applications seem boundless, promising a future where materials and technologies are designed with atomic precision to address humanity's most pressing challenges, from sustainable energy to complex disease treatment. The study of
electron configurations, therefore, isn't merely an academic pursuit; it's a vital investment in a future shaped by innovation, sustainability, and a deeper appreciation for the elegant order underlying the universe. It represents a continuous cycle of discovery – observing phenomena, formulating theories about electron behavior, developing technologies based on those theories, and then using those technologies to observe even more complex phenomena, leading to further refinement of our understanding.
Looking ahead, several key areas of research promise to further revolutionize our understanding and application of electron configurations. The development of advanced spectroscopic techniques, such as time-resolved core-level spectroscopy, will allow us to observe electron dynamics in real-time, providing unprecedented insights into chemical reactions and material transformations. Simultaneously, the rise of artificial intelligence and machine learning offers powerful tools for analyzing vast datasets of experimental and theoretical results, accelerating the discovery of novel materials with tailored electron configurations. Furthermore, the convergence of quantum computing with materials science holds the potential to simulate electron behavior with unparalleled accuracy, enabling the design of materials with properties currently beyond our reach.
Beyond the purely technological, the continued exploration of electron configurations in extreme environments – within neutron stars, for example, or under the intense pressures of planetary interiors – will undoubtedly challenge and refine our fundamental understanding of physics. These investigations will push the boundaries of our theoretical models and potentially reveal new states of matter and novel physical phenomena. The interplay between theoretical predictions and experimental observations in these extreme regimes will be crucial for validating our understanding of the universe at its most fundamental level. Ultimately, the ongoing quest to understand and harness the power of electron configurations is a testament to human curiosity and our relentless pursuit of knowledge, a journey that promises to unlock a future brimming with possibilities.